Hunt for the Killer Bug
<br>
The bacterium that causes Legionnaires’ disease remains difficult to track. French researchers have now developed a new technique that should allow living representatives of this dangerous pathogen to be detected much more quickly than with conventional methods.
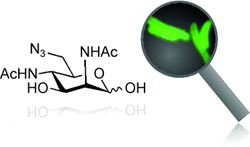
As they report in the journal Angewandte Chemie, samples are exposed to an azide-modified compound that the pathogen specifically incorporates into its shell, which is made of saccharide units. A fluorescent marker attached to the azide groups is used to identify the pathogen.
In the summer of 1976, a previously unknown disease broke out at a convention of the American Legion in Philadelphia. Of the 221 people infected, 34 died. The disease now known as Legionnaires’ disease has broken out many times since then.
The pathogen behind it was identified as a bacterium called Legionella pneumophila that proliferates in systems of standing water at temperatures between 25 and 50 °C, such as water reservoirs, boilers, fountains, whirlpool tubs, or intermittently used water pipes. Although drinking the contaminated water poses no risk, inhaling droplets leads to severe lung infections.
In order to avoid epidemics, it is necessary to monitor vulnerable systems. However, traditional testing methods based on bacterial cultures require 10 days to identify the pathogen – far too late to intervene in suspected cases.
Sam Dukan, Boris Vauzeilles, and their team at the Institute for the Chemistry of Natural Substances (CNRS, Gif-sur-Yvette), the Institut de Chimie Moléculaire et des Matériaux d’Orsay (CNRS/Université Paris-Sud) and the Institut de Microbiologie de la Méditérranée (CNRS/Aix-Marseille University) have now developed a new method that can be used to identify living bacteria of the species Legionella pneumophila within just one day.
Legionella are Gram-negative bacteria with a species-specific pattern of saccharide molecules on their surface, called the lipopolysaccharides. In Legionella pneumophila, these contain a special saccharide building block that other bacteria do not have. For their new test, the researchers exposed the sample to a precursor molecule of this saccharide with an additional azide group (–N3) tacked on. If the bacteria in question are present, they take up this substance and use it to build the saccharide building block to use in their lipopolysaccharides—which are now tagged with azide groups.
These groups can then be used to attach various probes to the surface of the cell. For example with a fluorescent label, the marked bacteria give off a green glow under a microscope. Because only Legionella pneumophila synthesizes the special saccharide building blocks, other species of Legionella are not marked in this process. This new method is the first successful metabolic lipopolysaccharide label using a specific saccharide that can selectively detect one species.
About the Author
Dr. Boris Vauzeilles is a French CNRS researcher with a dual appointment at the Institut de Chimie des Substances Naturelles in Gif-sur-Yvette and the Institut de Chimie Moléculaire in Orsay. His research interests are mainly focused on the design and synthesis of new tools for chemical biology, with a major expertise in glycochemistry. His work is done in close collaboration with biologists such as Dr. Sam Dukan.
Author: Boris Vauzeilles, Institut de Chimie des Substances Naturelles du CNRS, Gif-sur-Yvette (France), mailto:boris.vauzeilles@cnrs.fr
Title: Identification of Living Legionella pneumophila Using Species-Specific Metabolic Lipopolysaccharide Labeling
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201309072
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















