Enzyme designers: Simulation of the AsqJ enzyme opens up new options for pharmaceutical chemistry
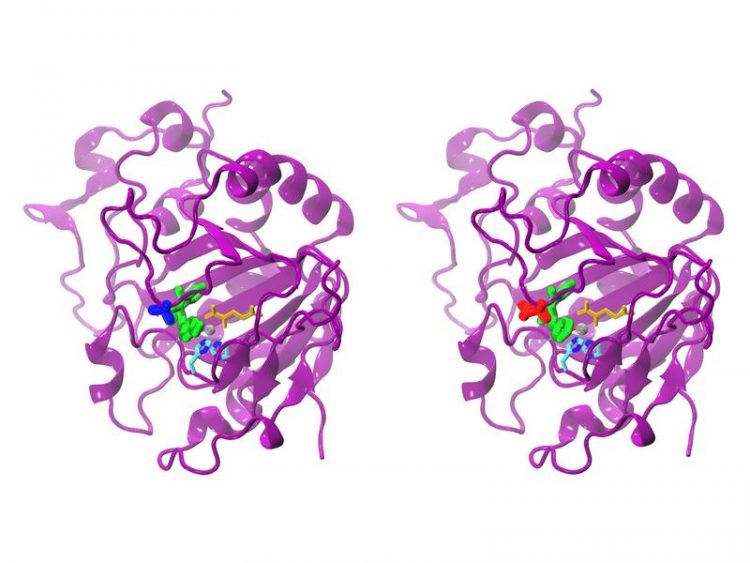
Natural (left) and modified AsqJ. While a valine is incorporated at position 72 in the natural (blue) enzyme, the modified form possesses an isoleucine (red) at position 72. Sophie Mader / TUM
Without enzymes, nature would come to a standstill. These tiny molecules accelerate biochemical reactions or make them possible in the first place. But how does this happen on a molecular level? “Understanding the exact function of enzymes is one of the greatest challenges of modern biochemistry,” says Ville Kaila, Professor of Computational Biocatalysis at the Technical University of Munich.
The research team led by Ville Kaila and Michael Groll, Professor of Biochemistry at the Technical University of Munich, have, for the first time, deciphered the mechanism of the enzyme aspoquinolone J (AsqJ), a dioxygenase that activates carbon bonds with oxygen.
One enzyme – many reactions
The enzyme AsqJ is particularly exciting as it catalyzes a cascade of chemical reactions that ultimately lead to the formation of antibacterial compounds. It was discovered only a few years ago in the Aspergillus nidulans fungus.
The researchers combined different methods to uncover the secrets held within the enzyme: First, Alois Bräuer and Prof. Michael Groll used X-ray crystallography to determine the three-dimensional atomic structure of the molecule. Sophie Mader and Ville Kaila then used this information to carry out quantum mechanical simulations on its biochemical processes.
Elucidating the secrets of AsqJ with simulations
“Our calculations illustrate how the enzyme catalyzes the formation of quinolone alkaloid,” reports Kaila. “Tiny details have amazing effects: A slight change in the substrate, like the removal of a small chemical group, is sufficient to practically stop the reaction.”
Next, the team computationally designed a new variant of the enzyme that catalyzes the formation of quinolone alkaloids with the modified substrate. This new enzyme was experimentally produced in bacteria and tested for its functionality. “The results were impressive: the expected reaction took place after only a few seconds,” recalls Bräuer.
Computational design of new compounds
“This experiment demonstrates that our methodology works and is also suited to represent the functionality of other enzymes at the molecular level,” says Ville Kaila. Enzyme design is still at a basic level, but it has enormous potential. In the future, we could aim to computationally design medical drugs, for example.
“The work demonstrates that our methodology is accurate and also well suited to study the functionality of other enzymes at the molecular level,” says Ville Kaila. Enzyme design is still basic research – but it has enormous potential. An aim of future research will be to design enzymes in a computer to, for example, produce new drugs.
Publication:
S. L. Mader, A. Bräuer, M. Groll, V. R. I. Kaila
Catalytic mechanism and molecular engineering of quinolone biosynthesis in dioxygenase AsqJ. Nature Communications 9(1), 1168 (2018) – DOI: 10.1038/s41467-018-03442-2
Further information:
The research was funded by the European Research Council (ERC), the German Research Foundation (DFG), the Collaborative Research Centers SFB 1035 and SFB 749 and the Cluster of Excellence Integrated Protein Science Munich (CIPSM). The project received further support as part of the research cooperation between the TUM and the King Abdullah University of Science and Technology (KAUST). The crystal structures were determined in cooperation with the synchrotron source of the Paul Scherrer Institute in Villigen (Switzerland). The computer simulations were carried out in cooperation with the Leibniz Computer Center of the Bavarian Academy of Sciences.
Contact:
Prof. Ville R. I. Kaila
Technical University of Munich
Professorship of Computational Biocatalysis
Tel.: +49 89 289 13612
E-Mail: ville.kaila@ch.tum.de
http://www.compbio.ch.tum.de/ Website of Prof. Kaila’s group
http://www.biochemie.ch.tum.de/ Website of Prof. Groll’s group
https://www.nature.com/articles/s41467-018-03442-2 Original publication
https://www.tum.de/nc/en/about-tum/news/press-releases/detail/article/34577/ Press release at tum.de
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















