Cellular Valve Structure Opens Up Potential Novel Therapies
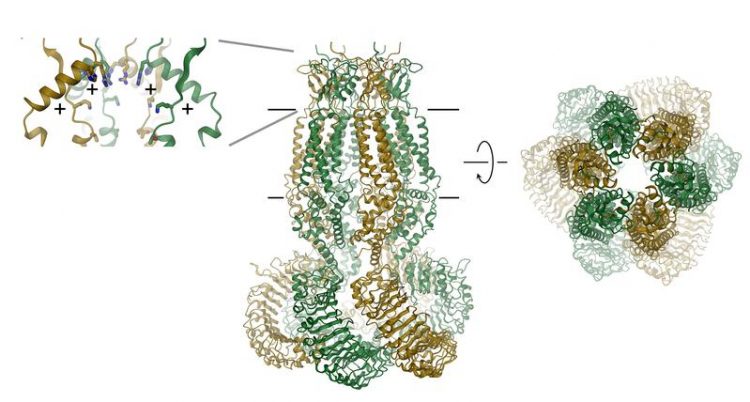
Structure of a volume-regulated chloride channel (center: ribbon diagram, right: selectivity filter, left: regions with positively charged amino acids) Raimund Dutzler, UZH
Human cells are enclosed by membranes and are in osmotic equilibrium with their environment. If the concentration of solute molecules (osmolarity) in the fluid surrounding the cells decreases, cells start to swell; in extreme cases, this can result in the cells bursting.
To avoid this, cells activate volume-regulated chloride channels (VRACs) of the LRRC8 protein family. If the cell volume increases as a result of inflowing water, these cellular valves open to allow the negatively charged chloride ions and uncharged osmolytes to flow out, returning the cell to its original state.
Structure of a volume-regulated anion channel
Although discovered only five years ago, important properties of these cellular valves have already been described. For example, it is known that, besides their role in volume regulation, VRACs play an important role for the uptake of drugs used in cancer therapy and that they are responsible for the uncontrolled release of neurotransmitters after a stroke.
Despite these advances, the molecular make-up of VRACs and the basis for their selectivity has remained elusive. Researchers at the Department of Biochemistry of the University of Zurich have now closed this gap in our understanding. Using cryo-electron microscopy and X-ray crystallography, the team led by Prof. Raimund Dutzler has determined the detailed molecular structure of a VRAC. In addition, the researchers analyzed the functional properties of the protein through electrophysiological techniques.
From form to function
VRACs consist of six subunits, which are arranged around an axis that defines the ion permeation pore. The protein, which is located in the membrane, contains a small extracellular and a large intracellular domain. The latter probably plays an important role in channel activation.
The extracellular domain constricts the channel and functions as selectivity filter. “The positive residues in this filter attract negatively charged chloride ions and allow their permeation while excluding larger molecules from entering the cell,” explains Raimund Dutzler.
Potential approaches for the treatment of ischemia and cancer
With their work, the UZH scientists have provided a foundation for a better understanding of the molecular mechanisms underlying cell volume control. “This knowledge provides a valuable basis for the development of potential novel drugs,” states Dutzler. In case of cerebral ischemia or stroke, the astrocytes in the brain swell.
The resulting uncontrolled outflow of the neurotransmitter glutamate, which is mediated by VRACs, has detrimental consequences for affected people. Such cases could benefit from the development of specific blockers. Another potential application relates to the role of VRACs in cancer therapy: A cell-specific activation of VRACs could improve the uptake of therapeutics into cancer cells.
Literature:
Dawid Deneka, Marta Sawicka, Andy K. M. Lam, Cristina Paulino and Raimund Dutzler. Structure of a volume-regulated anion channel of the LRRC8 family. Nature. May 16, 2018. DOI: 10.1038/s41586-018-0134-y
Project funding
The project was funded by a grant of the Swiss National Science Foundation (SNSF). The cryo-EM data were collected with electron microscopes of the Center for Microscopy and Image Analysis of UZH, which were acquired thanks to a substantial contribution of the Mäxi Foundation. X-ray data were collected at the Swiss Synchrotron Light Source of the Paul Scherrer Institute.
Contact:
Prof. Raimund Dutzler, PhD
Department of Biochemistry
University of Zurich
Phone: +41 44 635 65 50
E-mail: dutzler@bioc.uzh.ch
http://www.media.uzh.ch/en/Press-Releases/2018/volume-regulated-chloride-channel…
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















