Cells are crawling all over our bodies, but how?
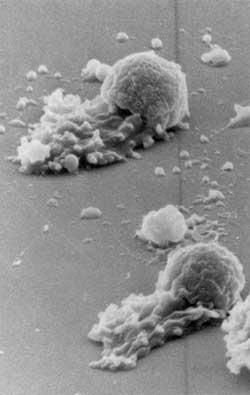
This is an electron microscope image of two crawling worm sperm magnified ~5,000X. Credit: Courtesy, Tom Roberts, FSU Dept. of Biological Science<br>
For better and for worse, human health depends on a cell's motility –– the ability to crawl from place to place. In every human body, millions of cells –are crawling around doing mostly good deeds ––– though if any of those crawlers are cancerous, watch out.
“This is not some horrible sci-fi movie come true but, instead, normal cells carrying out their daily duties,” said Florida State University cell biologist Tom Roberts. For 35 years he has studied the mechanical and molecular means by which amorphous single cells purposefully propel themselves throughout the body in amoeboid-like fashion ––absent muscles, bones or brains.
Meanwhile, human cells don't give up their secrets easily. In the body, they use the millions of tiny filaments found on their front ends to push the front of their cytoskeletons forward. In rapid succession the cells then retract their rears in a smooth, coordinated extension-contraction manner that puts inchworms to shame. Yet take them out of the body and put them under a microscope and the crawling changes or stops.
But now Roberts and his research team have found a novel way around uncooperative human cells.
In a landmark study led by Roberts and conducted in large part by his then-FSU postdoctoral associate Katsuya Shimabukuro, researchers used worm sperm to replicate cell motility in vitro –– in this case, on a microscope slide.
Doing what no other scientists had ever successfully done before, Shimabukuro disassembled and reconstituted a worm sperm cell, then devised conditions to promote thecell's natural pull-push crawling motions even in the unnatural conditions of a laboratory. Once launched, the reconstituted machinery moved just like regular worm sperm do in a natural setting –– giving scientists an unprecedented opportunity to watch it move.
Roberts called his former postdoc's signal achievement “careful, clever work” –– and work it did, making possible new, revealing images of cell motility that should help to pinpoint with never-before-seen precision just how cells crawl.
“Understanding how cells crawl is a big deal,” Roberts said. “The first line of defense against invading microorganisms, the remodeling of bones, healing wounds in the skin and reconnecting of neuronal circuits during regeneration of the nervous system –– all depend on the capacity of specialized cells to crawl.
“On the downside, the ability of tumor cells to crawl around is a contributing factor in the metastasis of malignancies,” he said. “But we believe our achievements in this latest round of basic research could eventually aid in the development of therapies that target cell motility in order to interfere with or block the metastasis of cancer.”
Funding for Robert's worm-sperm study came from the National Institutes of Health. The findings are described in a paper (“Reconstitution of Amoeboid Motility In Vitro Identifies a Motor-Independent Mechanism for Cell Body Retraction”) published online in the journal Current Biology.
Why worm sperm?
For one thing, said Roberts, the worm sperm is different from most cells in that itdoesn't use molecular motor proteins to facilitate its contractions; it shimmies along strictly by putting together and tearing down its tiny filaments. And the simple worm sperm makes a good model because, while it is similar to a human cell it has fewer moving parts, making it less complicated to take apart and reassemble than, say, brain or cancer cells.
Armed with the newfound ability to reconstitute amoeboid motility in vitro, cell biologists such as Roberts may be able to learn the answers to some major moving questions. Among them: How can some cells continue to crawl even after researchers have disabled their supply of myosin, the force-producing “mover protein” that functions like a motor to help power muscle and cell contraction?
For Roberts and his team, the next move will be to determine if what they've learned about worm sperm also applies to more conventional crawling cells, including tumor cells.
“As always, there will be more questions,” Roberts said. “Are there multiple mechanisms collaborating to drive cell body retraction? Is there redundancy built into the motility systems?”
Co-authors of the Current Biology paper include Roberts, a professor in the FSU Department of Biological Science; Shimabukuro, a former FSU postdoctoral associate in biology who now is a research scientist at the Japan Science and Technology Agency; Naoki Noda, of the Marine Biological Laboratory at Woods Hole, Mass.; and Murray Stewart, of the Medical Research Council's Laboratory of Molecular Biology in Cambridge, England.
Media Contact
More Information:
http://www.fsu.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















