Antibodies as ‘messengers’ in the nervous system
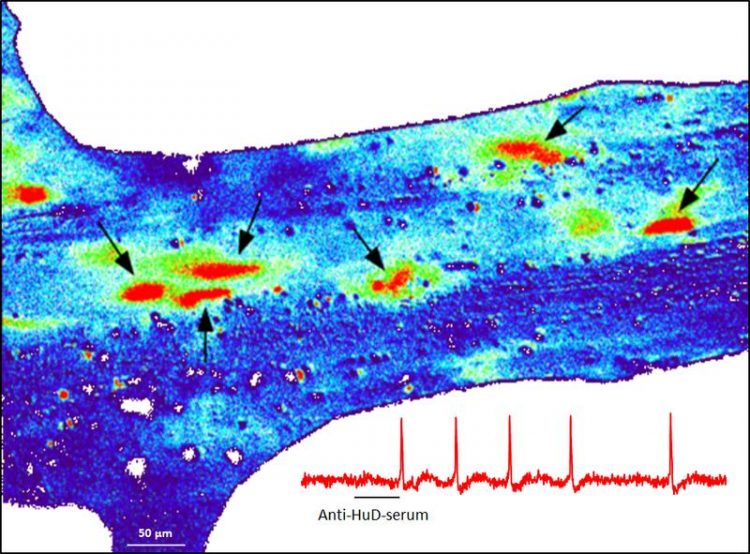
A Ganglion in the human intestine, which shows nerval activity after giving the anti-HuD-serum. The activity is red. (Fig.: Schemann, Michel/ TUM)
Functional disorders in organs that manifest in conjunction with tumors are called paraneoplastic syndromes. These syndromes are not caused by the primary tumor itself, but are instead frequently a result of the body’s autoimmune reaction. In such cases, a person’s own antibodies turn against their own cells and attack them.
One of these functional disorders is paralysis of the intestinal tract, for example intestinal pseudoobstruction. It makes it difficult for patients to obtain the nutrients and calories they require from their diet. The so-called anti-Hu syndrome is a type of paraneoplastic syndrome often associated with atonic gut and generally occurs in conjunction with small-cell lung cancer. Paraneoplastic syndromes often occur before the tumor is even detected.
Hu proteins are usually located in the nucleus of all nerve cells and consist of four family members (HuA, B, C, and D). Because the tumor releases the Hu protein, the immune system generates antibodies to fight it. Initially, they serve to defend against the tumor: The greater the concentration of antibodies, the slower the tumor grows. However, these anti-Hu antibodies — named after the first patient in whom these antibodies were discovered in 1985 — also result in an autoimmune reaction with severe gut disorders as an accompanying illness.
Nerves are activated before they can be damaged
Professor Michael Schemann and his colleagues at the Chair for Human Biology at TUM wanted to identify causes for possible nervous function disorders that occur in paraneoplastic syndromes and paralytic intestine. For this purpose, they examined serums from patients with small-cell lung cancer from the Mayo Clinic in Rochester, MN (USA). In a study conducted over a period of ten years, the researchers were able to show for the first time that these patient serums activate human nerve cells within milliseconds without causing neuronal damage. This modifies nerve functions long before the autoimmune reaction damages the nerves.
Working together with the company Euroimmun from Lübeck, the team was even able to identify the factor responsible for this: Normally, nerve cells are activated or inhibited via neurotransmitters that bind to specific structures in the cell membrane (receptors). Surprisingly, it turned out to be an antibody — namely the anti-HuD antibody — which stimulated the nerve cells in the patient serums.
Antibody mimics neurotransmitters acetylcholine and adenosine triphosphate
What was striking about this finding was the fact that the antibody does not achieve this effect binding to its genuine target protein. “Interestingly, the nerve-activating effect is transmitted via receptors for neurotransmitters,” said Professor Schemann. “These receptors are usually activated by acetylcholine and adenosine triphosphate.” In a nutshell, the antibody more or less mimics the effects of the neurotransmitters acetylcholine and adenosine triphosphate.
The HuD protein typically stabilizes ribonucleic acid (RNA) and has nothing to do with nerve activation. How and where exactly the anti-HuD antibody binds to the receptors continues to remain a black box. However, this newly discovered effect of the anti-HuD-antibody heralds a paradigm shift, according to Professor Schemann, because antibodies are able to activate nerves regardless of antibody-specific binding structures on the cell membrane.
“Although what we have found will not heal lung cancer itself,” Professor Schemann explained, “it will lead to new clinical understanding and hence hopefully to new therapeutic approaches for related paraneoplastic syndromes such as intestinal pseudoobstruction.”
Just recently, the research group at the Chair for Human Biology, in collaboration with the Charité in Berlin, demonstrated that antibodies are able to activate human nerves*. In this case, the functional principle was obvious, as the binding of the antibody to defined structures of a potassium channel modified the excitability of the nerves.
Publications:
Qin Li**, Klaus Michel**, Anita Annahazi, Ihsan E. Demir, Güralp O. Ceyhan, Florian Zeller, Lars Komorowski, Winfried Stöcker, Michael J. Beyak, David Grundy, Gianrico Farrugia, Roberto De Giorgio und Michael Schemann: Anti-Hu antibodies activate enteric and sensory neurons, Scientific Reports 12/2016. (** coordinate 1st-authors)
DOI: 10.1038/srep38216
http://www.nature.com/articles/srep38216
*Piepgras J, Höltje M, Michel K, Li Q, Otto C, Drenckhahn C, Probst C, Schemann M, Jarius S, Stöcker W, Balint B, Meinck HM, Buchert R, Dalmau J, Ahnert-Hilger G, Ruprecht K.: Anti-DPPX encephalitis, Neurology 2015 Sep 8;85(10):890-7.
DOI: 10.1212/WNL.0000000000001907
Contact:
Prof. Dr. Michael Schemann
Technical University of Munich
Chair for Human Biology
Tel: +49/8161/71 5403
schemann@wzw.tum.de
https://www.tum.de/die-tum/aktuelles/pressemitteilungen/detail/article/33691/
http://humanbiology.wzw.tum.de/index.php?id=24&L=1
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















