New agents to combat the plague
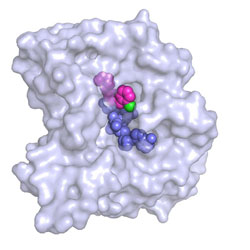
A newly developed inhibitor (magenta) from the pyridone group binds to a vital enzyme of the plague pathogen. The cofactor of the enzyme, NADH, which contributes significantly to the effect of the inhibitor, is shown in blue.<br>Image: Maria Hirschbeck<br>
A wonderful breakthrough for scientists from the universities of Würzburg and Stony Brook (USA): they have shed light on the binding and action mechanisms of two new inhibitors that attack the plague pathogen, the bacterium Yersinia pestis. In the current issue of the journal “Structure” they present both substances, which belong to the pyridone group.
The new inhibitors attach themselves to the bacterial enzyme FabV and impede it in its work. This enzyme performs the final step in the production of bacterial fatty acids. If it is blocked, the plague pathogen dies. This is because without fatty acids it cannot maintain its protective shell, the cell membrane.
“But the two substances do not inhibit the enzyme well enough yet,” says Professor Caroline Kisker of the Rudolf Virchow Center at the University of Würzburg. For that reason, these new inhibitors and their interactions with the enzyme now need to be analyzed further and improved.
Steps to improve the inhibitors
Structural biologist Kisker, her doctoral student Maria Hirschbeck, and postdoctoral fellow Jochen Kuper are collaborating on this with Würzburg chemists and pharmacists Professor Christoph Sotriffer and Steffen Wagner as well as with Peter Tonge of Stony Brook University. In their laboratories, these scientists are working not with pathogens of the plague, but with the isolated enzyme. They are crystallizing it as a compound with the inhibitors, rendering it into a state in which they can analyze how the inhibitors attach themselves to the enzyme right down to the molecular level.
Christoph Sotriffer, an expert in computer modeling of molecules, and his staff then examine the crystal structures and propose changes to the inhibitors that might make these even more effective. Finally, the modified inhibitors are synthesized and re-tested: test-tube experiments reveal whether they really are further weakening this enzyme that is so vital to the plague pathogen.
“This cycle will generally have to be repeated several times until, ideally, we end up with a highly active inhibitor,” says Caroline Kisker. However, whether the inhibitor will then also be suitable as medication is far from certain. Numerous other tests will be needed to determine that.
This research is contained within the Würzburg Collaborative Research Center 630 (Recognition, Preparation, and Functional Analysis of Agents against Infectious Diseases). The German Research Foundation (DFG) is funding the work.
Progression of an infection with the plague pathogen
The pathogens of the plague tend to live in rodents, particularly rats. They can be transferred to humans through bites from infected rat fleas. After one to seven days, the sufferer develops a high fever and shivering, among other things. These symptoms are joined by painful buboes, swellings of the lymph nodes that appear like lumps on the skin. In rare cases, the lumps rupture outwards, according to the Robert Koch Institute.
As the disease progresses, the pathogens may also attack internal organs, especially the lungs. It is then common for the sufferer to cough up blood. At this stage, the pathogens can also be transmitted from human to human in droplets of coughed-up fluid. Without treatment with antibiotics, this pneumonic plague, as it is known, almost always ends in death. With bubonic plague, on the other hand, there is a 50 percent chance of survival even without treatment. Generally speaking, if the plague is detected early, it can be treated successfully with antibiotics – provided that these are still effective.
Resistant plague pathogens discovered
In 2010, scientists from the Pasteur Institute in Paris found two plague pathogen strains that no longer respond to antibiotics. Both came from Madagascar. This island south-east of Africa is a hotspot for global outbreaks of the plague: in 2010, there were 313 recognized cases of the disease here, according to statistics from the World Health Organization (WHO). The second-highest incidence of infection was found in the Congo (152), followed by Peru with 27 cases.
“Structure of the Yersinia pestis FabV Enoyl-ACP Reductase and its Interaction with two 2-Pyridone Inhibitors”, Maria W. Hirschbeck, Jochen Kuper, Hao Lu, Nina Liu, Carla Neckles, Sonam Shah, Steffen Wagner, Christoph A. Sotriffer, Peter J. Tonge, and Caroline Kisker. Structure, Vol. 20, Issue 1, 89-100, 11 January 2012, DOI 10.1016/j.str.2011.07.019
Contact
Prof. Dr. Caroline Kisker, Institute of Structural Biology, Rudolf Virchow Center / DFG Research Center for Experimental Biomedicine, T +49 (0)931 31-80381, caroline.kisker@virchow.uni-wuerzburg.de
Media Contact
More Information:
http://www.virchow.uni-wuerzburg.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















