A molecular dance of phospholipid synthesis
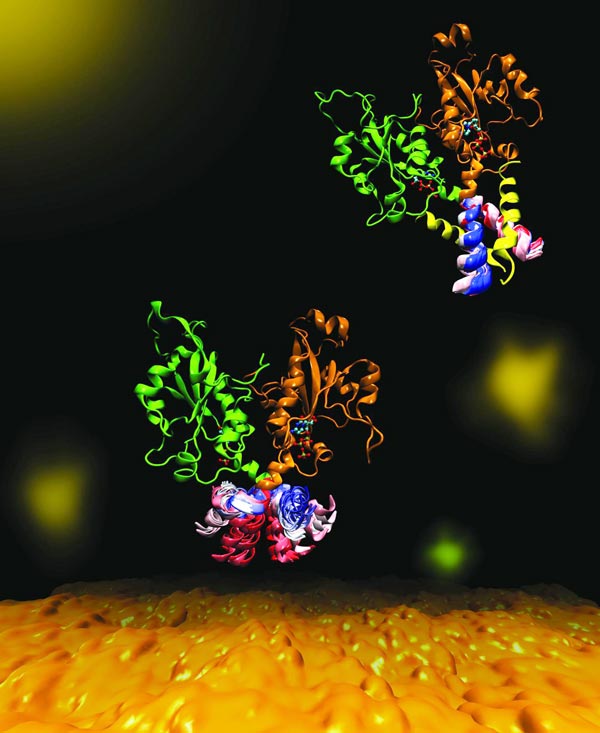
CCT is a key enzyme that maintains a balanced composition of cell membrane phospholipids. Image highlights the dynamics of a portion of the enzyme CCT that is essential for regulation of its functions. The molecular dynamics was explored in a collaboration between the Cornell and Tieleman labs using computational methods. Credit: Mohsen Ramezanpour and Jaeyong Lee
Rosemary Cornell, a professor of molecular biology and biochemistry at Simon Fraser University in Canada, studies the enzyme CTP:phosphocholine cytidylyltransferase, or CCT. CCT sets the rate of PC production in cells by binding to cell membranes with low PC content.
When bound to membranes, the CCT enzyme changes shape in a way that allows it to carry out the key rate-limiting step in PC synthesis. When the amount of PC making up the membrane increases, the CCT falls off the membrane, and PC production ceases.
“The membrane is this big macromolecular array with lots of different molecules in it,” Cornell said. “How does this enzyme recognize that 'Oh, I should slow down because the PC content of the membrane is getting too high?'”
Cornell and her project team – a collaboration with Peter Tieleman and graduate student, Mohsen Ramezanpour at the University of Calgary and Jaeyong Lee and Svetla Taneva, research associates at SFU – thought that the answer must have to do with the dynamic changes in shape that the enzyme undergoes when it binds to a membrane.
But these changes are difficult to capture with traditional structural biology methods like x-ray crystallography, which take a static snapshot of molecules. Instead, the team used computational simulations of molecular dynamics, which use information about the forces between every individual atom in a molecule to calculate the trajectories of the enzyme's moving parts.
“What it looks like (when you visualize the output) is your big molecule dancing in front of your eyes,” Cornell said. “We set up the molecular dynamics simulation not once, not twice, but 40 different (times). It took months and months just to do the computational parts and even more months trying to analyze the data afterward. We actually spent a lot of time once we got the data just looking on the screen at these dancing molecules.”
The simulated dance of the CCT molecule showed that when the M-domain, the section of the enzyme that typically binds to the membrane, detaches from a membrane, it snags the active site of the enzyme, preventing it from carrying out its reaction. When the snagging segment was removed from the simulation, the team saw a dramatic bending motion in the docking site for the snagging element, and speculated that this bending would create a better enzyme active site for catalyzing the reaction when attached to a membrane. The team confirmed these mechanisms using biochemical laboratory experiments.
Interestingly, previous genetic studies had shown that mutations in the gene encoding CCT are responsible for rare conditions like spondylometaphyseal dysplasia with cone-rod dystrophy, which causes severe impairments in bone growth and vision, but it was unknown how these changes in the enzyme could lead to such dramatic consequences. Cornell hopes that understanding how the enzyme works could help researchers find out.
“If you have just one small change in CCT, then how is that going to make this whole process of synthesizing PC defective?” Cornell asks. “That's what we're studying right now.”
###
The study was funded by the Canadian Institutes for Health Research.
About the Journal of Biological Chemistry
JBC is a weekly peer-reviewed scientific journal that publishes research “motivated by biology, enabled by chemistry” across all areas of biochemistry and molecular biology. The read the latest research in JBC, visit http://www.
About the American Society for Biochemistry and Molecular Biology
The ASBMB is a nonprofit scientific and educational organization with more than 12,000 members worldwide. Most members teach and conduct research at colleges and universities. Others conduct research in various government laboratories, at nonprofit research institutions and in industry. The Society's student members attend undergraduate or graduate institutions. For more information about ASBMB, visit http://www.
Media Contact
More Information:
http://dx.doi.org/10.1074/jbc.RA118.002053All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

“Nanostitches” enable lighter and tougher composite materials
In research that may lead to next-generation airplanes and spacecraft, MIT engineers used carbon nanotubes to prevent cracking in multilayered composites. To save on fuel and reduce aircraft emissions, engineers…

Trash to treasure
Researchers turn metal waste into catalyst for hydrogen. Scientists have found a way to transform metal waste into a highly efficient catalyst to make hydrogen from water, a discovery that…

Real-time detection of infectious disease viruses
… by searching for molecular fingerprinting. A research team consisting of Professor Kyoung-Duck Park and Taeyoung Moon and Huitae Joo, PhD candidates, from the Department of Physics at Pohang University…





















