U of M discovery to improve efficiencies in fuel, chemical and pharmaceutical industries

University of Minnesota engineering researchers are leading an international team that has made a major breakthrough in developing a catalyst used during chemical reactions in the production of gasoline, plastics, biofuels, pharmaceuticals, and other chemicals. The discovery could lead to major efficiencies and cost-savings in these multibillion-dollar industries.
The research is to be published in the June 29, 2012 issue of the leading scientific journal Science.
“The impact of this new discovery is enormous,” said the team's lead researcher Michael Tsapatsis, a chemical engineering and materials science professor in the University of Minnesota College of Science and Engineering. “Every drop of gasoline we use needs a catalyst to change the oil molecules into usable gasoline during the refining process.”
This research improves efficiencies by giving molecules fast access to the catalysts where the chemical reactions occur. Tsapatsis compared it to our use of freeways and side streets in our daily lives.
“It's faster and more efficient to use freeways to get where we want to go and exit to do our business compared to driving the side streets the entire way,” he explained. “The catalysts used today are more like all side streets. Molecules move slowly and get stuck. The efficiencies of these new catalysts could lower the costs of gasoline and other products for all of us.”
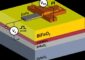
The research team built their prototype of the new catalyst using highly optimized ultra-thin zeolite nanosheets. They used a unique process to encourage growth of these nanosheets at 90-degree angles, similar to building a house of cards. The house-of-cards arrangement of the nanosheets makes the catalyst faster, more selective and more stable, but can be made at the same cost (or possibly cheaper) than traditional catalysts.
With faster catalysts available at no extra cost to the producer, production per manufacturing dollar will increase. With a higher output, it is conceivable that consumer costs will drop.
This new discovery builds upon previous discoveries at the University of Minnesota of ultra-thin zeolite nanosheets used as specialized molecular sieves for production of both renewable and fossil-based fuels and chemicals. These discoveries, licensed by the new Minnesota start-up company Argilex Technologies, are key components of the company's materials-based platform. The development of the new catalyst is complete, and the material is ready for customer testing.
“This breakthrough can have a major impact on both the conversion of natural gas to higher value chemicals and fuels, and on bio- and petroleum refiners,” said Cesar Gonzalez, CEO of Argilex Technologies. “Using catalysts made by this novel approach, refiners will be able to obtain a higher yield of desirable products such as gasoline, diesel, ethylene and propylene. At Argilex, we envision this catalyst technology platform to become a key contributor to efficient use of natural resources and improved economics of the world's largest industries.”
Researchers on the team are from around the globe. In addition to the University of Minnesota, researchers are from institutions in Tokyo, Abu Dhabi, Korea and Sweden.
Primary funding for this research is from the U.S. Department of Energy's Center for Catalysis and Energy Innovation, an Energy Frontier Research Center. The University of Minnesota is a partner in this multi-institutional research center at the University of Delaware. Other funding for this research is from the National Science Foundation Emerging Frontiers in Research and Innovation Program, the University of Minnesota's Initiative for Renewable Energy and the Environment, and the Abu Dhabi-Minnesota Institute for Research Excellence (ADMIRE) partnership between the University of Minnesota and the Abu Dhabi Petroleum Institute.
Read the full research paper entitled “Synthesis of Self-Pillared Zeolite Nanosheets by Repetitive Branching,” on the Science website: http://z.umn.edu/catalyst.
Media Contact
More Information:
http://www.umn.eduAll latest news from the category: Power and Electrical Engineering
This topic covers issues related to energy generation, conversion, transportation and consumption and how the industry is addressing the challenge of energy efficiency in general.
innovations-report provides in-depth and informative reports and articles on subjects ranging from wind energy, fuel cell technology, solar energy, geothermal energy, petroleum, gas, nuclear engineering, alternative energy and energy efficiency to fusion, hydrogen and superconductor technologies.
Newest articles

“Nanostitches” enable lighter and tougher composite materials
In research that may lead to next-generation airplanes and spacecraft, MIT engineers used carbon nanotubes to prevent cracking in multilayered composites. To save on fuel and reduce aircraft emissions, engineers…

Trash to treasure
Researchers turn metal waste into catalyst for hydrogen. Scientists have found a way to transform metal waste into a highly efficient catalyst to make hydrogen from water, a discovery that…

Real-time detection of infectious disease viruses
… by searching for molecular fingerprinting. A research team consisting of Professor Kyoung-Duck Park and Taeyoung Moon and Huitae Joo, PhD candidates, from the Department of Physics at Pohang University…