Solving fossil mystery could aid quest for ancient life on Mars
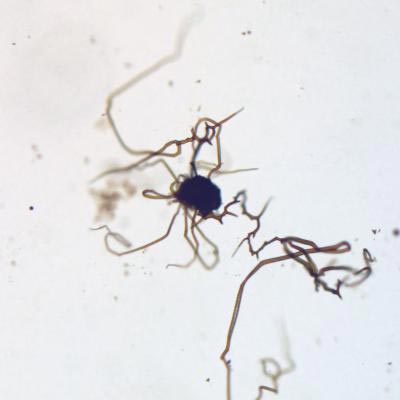
A life-like structure created in the lab by chemical gardening. Credit: Sean McMahon
Research which suggests that structures previously thought to be fossils may, in fact, be mineral deposits could save future Mars missions valuable time and resources.
Microscopic tubes and filaments that resemble the remains of tiny creatures may have been formed by chemical reactions involving iron-rich minerals, the study shows.
Previous research had suggested that such structures were among the oldest fossils on Earth.
The new findings could aid the search for extraterrestrial life during future missions to Mars by making it easier to distinguish between fossils and non-biological structures.
The discovery was made by a scientist from the University of Edinburgh who is developing techniques to seek evidence that life once existed on Mars.
Astrobiologist Sean McMahon created tiny formations in the lab that closely mimic the shape and chemical composition of iron-rich structures commonly found in Mars-like rocks on Earth, where some examples are thought to be around four billion years old.
Dr McMahon created the complex structures by mixing iron-rich particles with alkaline liquids containing the chemicals silicate or carbonate.
This process – known as chemical gardening – is thought to occur naturally where these chemicals abound. It can occur in hydrothermal vents on the seabed and when deep groundwater circulates through pores and fractures in rocks.
His findings suggest that structure alone is not sufficient to confirm whether or not microscopic life-like formations are fossils. More research will be needed to say exactly how they were formed.
The study, published in the journal Proceedings of the Royal Society B, was funded by the European Union's Horizon 2020 programme.
Dr Sean McMahon said: “Chemical reactions like these have been studied for hundreds of years but they had not previously been shown to mimic these tiny iron-rich structures inside rocks. These results call for a re-examination of many ancient real-world examples to see if they are more likely to be fossils or non-biological mineral deposits.”
Media Contact
All latest news from the category: Earth Sciences
Earth Sciences (also referred to as Geosciences), which deals with basic issues surrounding our planet, plays a vital role in the area of energy and raw materials supply.
Earth Sciences comprises subjects such as geology, geography, geological informatics, paleontology, mineralogy, petrography, crystallography, geophysics, geodesy, glaciology, cartography, photogrammetry, meteorology and seismology, early-warning systems, earthquake research and polar research.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…




















