Scientists make new estimates of the deep carbon cycle
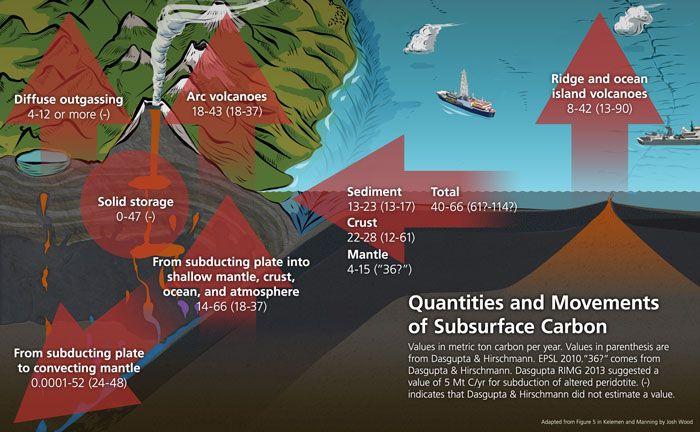
Major fluxes of carbon estimated by Craig Manning and Peter Kelemen. Courtesy of Josh Wood
Craig Manning, a professor of geology and geochemistry at UCLA, and Peter Kelemen, a geochemistry professor at Columbia University, present new analyses that represent an important advance in refining our understanding of Earth's deep carbon cycle.
Manning and Kelemen studied how carbon, the chemical basis of all known life, behaves in a variety of tectonic settings. They assessed, among other factors, how much carbon is added to Earth's crust and how much carbon is released into the atmosphere. The new model combines measurements, predictions and calculations.
Their research includes analysis of existing data on samples taken at sites around the world as well as new data from Oman.
The carbon 'budget' near the Earth's surface exerts important controls on global climate change and our energy resources, and has important implications for the origin and evolution of life, Manning said. Yet much more carbon is stored in the deep Earth. The surface carbon that is so important to us is made available chiefly by volcanic processes originating deep in the planet's interior.
Today carbon can return to Earth's deep interior only by subduction — the geologic process by which one tectonic plate moves under another tectonic plate and sinks into the Earth's mantle. Previous research suggested that roughly half of the carbon stored in subducted oceanic mantle, crust and sediments makes it into the deep mantle. Kelemen and Manning's new analysis suggests instead that subduction may return almost no carbon to the mantle, and that 'exchange between the deep interior and surface reservoirs is in balance.'
Some carbon must make it past subduction zones. Diamonds form in the mantle both from carbon that has never traveled to Earth's surface, known as primordial carbon, and from carbon that has cycled from the mantle to the surface and back again, known as recycled carbon. Manning and Kelemen corroborated their findings with a calculation based on the characteristics of diamonds, which form from carbon in the earth's mantle.
Deep carbon is important because the carbon at the Earth's surface, on which we depend, 'exists only by permission of the deep Earth,' Manning said, quoting a friend. At times in the Earth's history, the planet has been warmer (in the Cretaceous period, for example), and shallow seas covered North America. The new research sheds light on the Earth's climate over geologic time scales.
Media Contact
All latest news from the category: Earth Sciences
Earth Sciences (also referred to as Geosciences), which deals with basic issues surrounding our planet, plays a vital role in the area of energy and raw materials supply.
Earth Sciences comprises subjects such as geology, geography, geological informatics, paleontology, mineralogy, petrography, crystallography, geophysics, geodesy, glaciology, cartography, photogrammetry, meteorology and seismology, early-warning systems, earthquake research and polar research.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















