Superhydrophobic coating protects without the price
An environmentally friendly superhydrophobic coating repels water as effectively as commercial coatings that employ hazardous materials, according to scientists at Rice University and the University of Swansea. Credit: Shirin Alexander/University of Swansea
A new class of superhydrophobic nanomaterials might simplify the process of protecting surfaces from water.
A material made by scientists at Rice University, the University of Swansea, the University of Bristol and the University of Nice Sophia Antipolis is inexpensive, nontoxic and can be applied to a variety of surfaces via spray- or spin-coating.
The researchers led by Rice chemist Andrew Barron reported their find in the American Chemical Society journal ACS Applied Materials and Interfaces.
The hydrocarbon-based material may be a “green” replacement for costly, hazardous fluorocarbons commonly used for superhydrophobic applications, Barron said.
“Nature knows how to make these materials and stay environmentally friendly,” Barron said. “Our job has been to figure out how and why, and to emulate that.”
The lotus leaf was very much on their minds as the researchers tried to mimic one of the most hydrophobic — water-repelling — surfaces on the planet. Barron said the leaf's abilities spring from its hierarchy of microscopic and nanoscale double structures.
“In the lotus leaf, these are due to papillae within the epidermis and epicuticular waxes on top,” he said. “In our material, there is a microstructure created by the agglomeration of alumina nanoparticles mimicking the papillae and the hyperbranched organic moieties simulating the effect of the epicuticular waxes.”
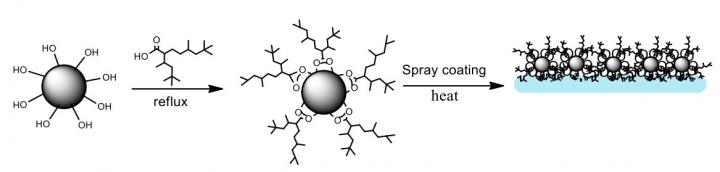
Fabrication and testing of what the researchers call a branched hydrocarbon low-surface energy material (LSEM) were carried out by lead author Shirin Alexander, a research officer at the Energy Safety Research Institute at the Swansea University Bay Campus.
There, Alexander coated easily synthesized aluminum oxide nanoparticles with modified carboxylic acids that feature highly branched hydrocarbon chains. These spiky chains are the first line of defense against water, making the surface rough. This roughness, a characteristic of hydrophobic materials, traps a layer of air and minimizes contact between the surface and water droplets, which allows them to slide off.
To be superhydrophobic, a material has to have a water contact angle larger than 150 degrees. Contact angle is the angle at which the surface of the water meets the surface of the material. The greater the beading, the higher the angle. An angle of 0 degrees is basically a puddle, while a maximum angle of 180 degrees defines a sphere just touching the surface.
The Barron team's LSEM, with an observed angle of about 155 degrees, is essentially equivalent to the best fluorocarbon-based superhydrophobic coatings, Barron said. Even with varied coating techniques and curing temperatures, the material retained its qualities, the researchers reported.
Potential applications include friction-reducing coatings for marine applications where there is international agreement in trying to keep water safe from such potentially dangerous additives as fluorocarbons, Barron said. “The textured surfaces of other superhydrophobic coatings are often damaged and thus reduce the hydrophobic nature,” he said. “Our material has a more random hierarchical structure that can sustain damage and maintain its effects.”
He said the team is working to improve the material's adhesion to various substrates, as well as looking at large-scale application to surfaces.
###
Co-authors of the paper are Julian Eastoe, a professor of chemistry at the University of Bristol; Alex Lord, a researcher at the Swansea University Center for Nanohealth; and Frédéric Guittard, a professor at the University of Nice Sophia Antipolis. Barron is the Charles W. Duncan Jr.-Welch Professor of Chemistry and a professor of materials science and nanoengineering at Rice and the Sêr Cymru Chair of Low Carbon Energy and Environment at Swansea.
The Robert A. Welch Foundation and the Welsh Government Sêr Cymru Program supported the research.
Read the abstract at http://pubs.
This news release can be found online at
http://news.
Follow Rice News and Media Relations via Twitter @RiceUNews
Video:
https:/
Related Materials:
Barron Research Group: http://barron.
Wiess School of Natural Sciences: http://naturalsciences.
Images for download:
http://news.
An environmentally friendly superhydrophobic coating repels water as effectively as commercial coatings that employ hazardous materials, according to scientists at Rice University and the University of Swansea. (Credit: Shirin Alexander/University of Swansea)
http://news.
A scanning electron microscope image of a new superhydrophobic material shows the rough surface of functionalized alumina nanoparticles. Scientists at Rice University and the University of Swansea led the creation of the environmentally friendly material. (Credit: University of Swansea)
Located on a 300-acre forested campus in Houston, Rice University is consistently ranked among the nation's top 20 universities by U.S. News & World Report. Rice has highly respected schools of Architecture, Business, Continuing Studies, Engineering, Humanities, Music, Natural Sciences and Social Sciences and is home to the Baker Institute for Public Policy. With 3,910 undergraduates and 2,809 graduate students, Rice's undergraduate student-to-faculty ratio is 6-to-1. Its residential college system builds close-knit communities and lifelong friendships, just one reason why Rice is ranked No. 1 for best quality of life and for lots of race/class interaction by the Princeton Review. Rice is also rated as a best value among private universities by Kiplinger's Personal Finance. To read “What they're saying about Rice,” go to http://tinyurl.
David Ruth
713-348-6327
david@rice.edu
Mike Williams
713-348-6728
mikewilliams@rice.edu
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

“Nanostitches” enable lighter and tougher composite materials
In research that may lead to next-generation airplanes and spacecraft, MIT engineers used carbon nanotubes to prevent cracking in multilayered composites. To save on fuel and reduce aircraft emissions, engineers…

Trash to treasure
Researchers turn metal waste into catalyst for hydrogen. Scientists have found a way to transform metal waste into a highly efficient catalyst to make hydrogen from water, a discovery that…

Real-time detection of infectious disease viruses
… by searching for molecular fingerprinting. A research team consisting of Professor Kyoung-Duck Park and Taeyoung Moon and Huitae Joo, PhD candidates, from the Department of Physics at Pohang University…