Extending a Battery's Lifetime with Heat

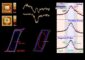
Asghar Aryanfar Naked-eye observation of amorphous/fractal lithium dendrites.
Don't go sticking your electronic devices in a toaster oven just yet, but for a longer-lasting battery, you might someday heat them up when not in use. Over time, the electrodes inside a rechargeable battery cell can grow tiny, branch-like filaments called dendrites, causing short circuits that kill the battery or even ignite it in flames.
But thanks to new experiments and computer simulations, researchers from the California Institute of Technology have explored in detail how higher temperatures can break down these dendrites — and possibly extend battery lifetimes.
A battery cell consists of a positive and negative electrode, called the cathode and anode. As the battery produces electrical current, electrons flow from the anode through a circuit outside the battery and back into the cathode. Having lost the electrons that are generating the current, some of the atoms in the anode — an electrically conductive metal like lithium — become ions that then travel to the cathode, moving through a conductive liquid medium called an electrolyte.
Recharging the battery reverses the process, and the ions travel back and stick onto the anode. But when they do, the ions don't attach evenly. Instead, they form microscopic bumps that eventually grow into long branches after multiple recharging cycles. When these dendrites reach and contact the cathode, they form a short circuit. Electrical current now flows across the dendrites instead of the external circuit, rendering the battery useless and dead.
The current also heats up the dendrites, and because the electrolyte tends to be flammable, the dendrites can ignite. Even if the dendrites don't short circuit the battery, they can break off from the anode entirely and float around in the electrolyte. In this way, the anode loses material, and the battery can't store as much energy.
“Dendrites are hazardous and reduce the capacity of rechargeable batteries,” said Asghar Aryanfar, a scientist at Caltech, who led the new study that's published this week on the cover of The Journal of Chemical Physics, from AIP Publishing. Although the researchers looked at lithium batteries, which are among the most efficient kind, their results can be applied broadly. “The dendrite problem is general to all rechargeable batteries,” he said.
The researchers grew lithium dendrites on a test battery and heated them over a couple days. They found that temperatures up to 55 degrees Celsius shortened the dendrites by as much as 36 percent. To figure out what exactly caused this shrinkage, the researchers used a computer to simulate the effect of heat on the individual lithium atoms that comprise a dendrite, which was modeled with the simple, idealized geometry of a pyramid.
The simulations showed that increased temperatures triggered the atoms to move around in two ways. The atom at the tip of the pyramid can drop to lower levels. Or, an atom at a lower level can move and leave behind a vacant spot, which is then filled by another atom. The atoms shuffle around, generating enough motion to topple the dendrite.
By quantifying how much energy is needed to change the structure of the dendrite, Aryanfar said, researchers can better understand its structural characteristics. And while many factors affect a battery's longevity at high temperatures — such as its tendency to discharge on its own or the occurrence of other chemical reactions on the side — this new work shows that to revitalize a battery, all you might need is some extra heat.
The article, “Annealing kinetics of electrodeposited lithium dendrites,” is authored by Asghar Aryanfar, Tao Cheng, Agustin J. Colussi, Boris V. Merinov, William A. Goddard III and Michael R. Hoffmann. It will be published in The Journal of Chemical Physics on October 1, 2015 (DOI: 10.1063/1.4930014). After that date, it can be accessed at: http://scitation.aip.org/content/aip/journal/jcp/143/13/10.1063/1.4930014
The authors of this study are affiliated with the California Institute of Technology.
ABOUT THE JOURNAL
The Journal of Chemical Physics publishes concise and definitive reports of significant research in the methods and applications of chemical physics. See: http://jcp.aip.org
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…