Rift Valley fever virus: An infection mechanism identified
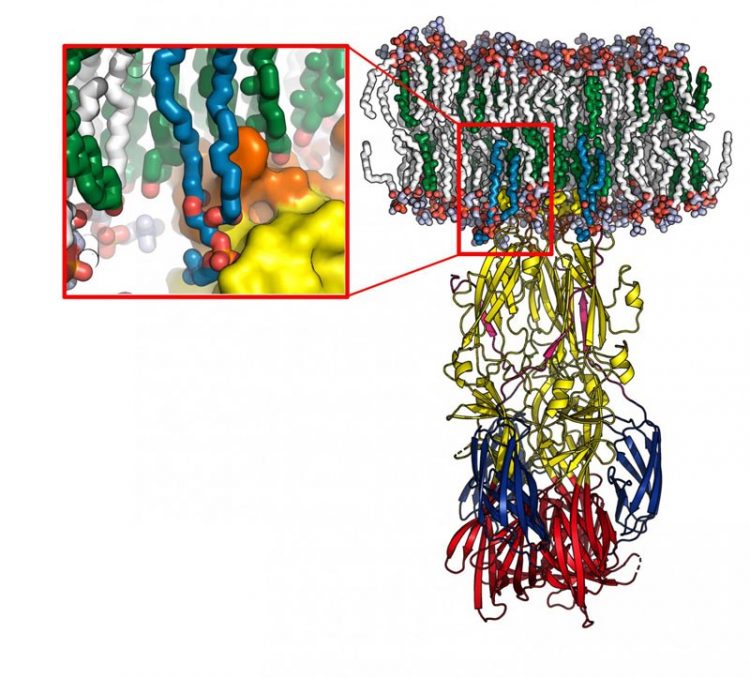
Model of the Rift Valley virus fusion protein interacting with a cell membrane (in white and green), obtained using molecular dynamics. The enlargement shows the lipid head 'recognition pocket', with the protein represented at the surface. Credit: © Pablo Guardado-Calvo, Institut Pasteur
They have demonstrated that the viral envelope protein has a “pocket” that specifically recognizes a category of lipids in the cell membrane. This pocket is also found in other human pathogenic viruses transmitted by different mosquitoes, such as the Zika and chikungunya viruses.
Understanding these interactions should pave the way for the identification of new therapeutic strategies that target viruses transmitted by these mosquitoes. The findings were published in the journal Science on November 3, 2017.
Rift Valley fever (RVF) virus is a bunyavirus that was first isolated from sheep in Kenya in 1930. The spread of RVF has serious economic consequences in Africa. The virus also causes severe disease in humans who come into contact with contaminated animals or who are bitten by infected mosquitoes, resulting in severe encephalitis and hemorrhagic fever that can prove fatal.
RVF therefore also represents a significant public health threat. In 2000, the virus spread outside the African continent to Saudi Arabia and Yemen. There are concerns that it may also extend to Asia and Europe.
RVF virus spreads in its host by fusing with cell membranes so that it can proliferate and infect other cells. Scientists in the Structural Virology Unit (Institut Pasteur/CNRS) directed by Félix Rey, in collaboration with the University of Göttingen, characterized the mechanism used by the virus to insert one of its surface proteins into the host cell membrane and drive fusion.
They also determined the atomic structure of this new protein-lipid complex, demonstrating that this protein has a “pocket” which specifically recognizes the hydrophilic heads of some of the lipids that make up the cell membrane. Importantly, this “recognition pocket” is found not only in RVF virus but also in the envelope proteins of other viral families transmitted by arthropods, such as the dengue, Zika and chikungunya viruses, which have caused major worldwide epidemics in recent years.
In the homologous protein of the chikungunya virus, the scientists pinpointed one of the residues of the recognition pocket as amino acid 226. In 2006, the A226V mutation enabled chikungunya to be transmitted by a new species of mosquito that is prevalent on Reunion Island (Aedes albopictus, or the tiger mosquito).
“This study offers a further illustration of the power of comparative analyses of viruses that appear very distant, such as bunyaviruses, alphaviruses and flaviviruses, which can result in highly significant findings and reveal shared mechanisms of action,” commented Félix Rey, Head of the Structural Virology Unit (Institut Pasteur/CNRS), where the study was carried out.
Understanding the mechanism used by these viruses for insertion in the cell membrane paves the way for the development of therapeutic agents that target the “pocket” involved in the fusion of viral and cell membranes with the aim of preventing pathogenic arboviruses from entering host cells.
Media Contact
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Security vulnerability in browser interface
… allows computer access via graphics card. Researchers at Graz University of Technology were successful with three different side-channel attacks on graphics cards via the WebGPU browser interface. The attacks…

A closer look at mechanochemistry
Ferdi Schüth and his team at the Max Planck Institut für Kohlenforschung in Mülheim/Germany have been studying the phenomena of mechanochemistry for several years. But what actually happens at the…

Severe Vulnerabilities Discovered in Software to Protect Internet Routing
A research team from the National Research Center for Applied Cybersecurity ATHENE led by Prof. Dr. Haya Schulmann has uncovered 18 vulnerabilities in crucial software components of Resource Public Key…