Newly discovered mechanism regulates myocardial distensibility
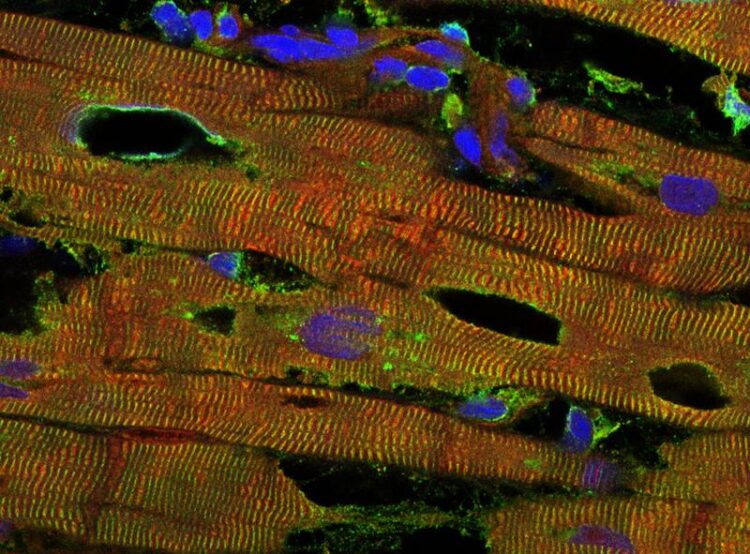
Immunofluorescence staining of the muscle tissue of a chronically diseased human heart under the confocal microscope.
Image: Linke Lab
A team of researchers headed by Münster University physiologist Prof. Wolfgang Linke has shown that oxidative stress, in combination with the extension of the heart walls, triggers a change in cardiac stiffness. A key role is played by the giant protein titin. This newly discovered mechanism is relevant, for example, in cases of an acute heart attack or chronic heart disease. The results have been published in the journal “PNAS”.
A healthy heart beats 50 to 100 times a minute and pumps 8,000 litres of blood around our body every day. A precondition for this function is the elasticity of the cardiac walls, which dilate as blood flows in (diastole) and contract again as the blood flows out (systole). Millions of tiny cavities in the heart muscle fibres, the sarcomeres, are responsible for this movement. These contractile units contain the largest protein found in the human body called titin. It has the function of a mechanical spring, which develops a restoring force during the extension of the sarcomeres – just like a rubber band.
Working with researchers from the Universities of Bochum, Würzburg, Cologne, Regensburg, Göttingen and Düsseldorf, a team of researchers at the University of Münster headed by Prof. Wolfgang Linke, Director of the Institute of Physiology II, has now discovered that oxidative stress, in combination with the stretching of the heart walls, triggers a change in cardiac stiffness. The elastic titin in the sarcomeres is more strongly oxidised and, as a result, is modulated in its extensibility. The researchers call this newly discovered mechanism UnDOx (“Unfolded Domain Oxidation”). The study has been published in the journal “PNAS”.
Background and method
In the human organism, titin forms the backbone of the sarcomeres, the smallest functional units of the skeletal muscles and the heart. Titin provides both stability and elasticity to muscle cells, due its unique structure. For the first time, the research team has now shown in heart tissue of mice that oxidative stress together with the extension of the heart modulates the spring function of titin. Oxidative stress occurs when too many reactive oxygen species are present in the cell of an organism. These oxygen species, which include so-called free radicals, can cause cell damage. In small quantities, however, they regulate important physiological functions.
The researchers used a mass spectrometer to determine the oxidation status of the proteins in the heart, including titin. Additionally, they isolated heart muscle cells from deep-frozen tissue of a human heart and attached them to a force sensor and a micromotor, in order to stretch the specimens. This enabled them to measure the forces generated upon stretch and observe their increase or decrease during different forms of oxidative stress. In addition, the team produced recombinant titin molecules and also mutated them in a test tube, such that oxidation could no longer occur. “We then used a so-called atomic force microscope to directly measure the effects of stretch and oxidation on the titin spring,” Wolfgang Linke explains. “Using this device, we were able to stretch individual titin molecules like a rubber band and record the force upon extension, as well as the changes in force in the presence of an oxidant.”
The researchers demonstrated in their experiments that the UnDOx mechanism occurs in hearts under oxidative stress. This is the case, for example, after an acute heart attack or in chronic heart disease associated with altered cardiac filling. “The mechanism regulates the distensibility of the heart,” Linke adds. “Extensive wall stiffness is not good for the heart, because less blood flows in. In the case of diastolic heart failure, which is often found in elderly people, the stiffening of the heart is a major problem. We hope that such hearts can be made more distensible through the pharmacological regulation of titin oxidation, i.e. by using medication.”
The work was funded by the German Research Foundation (DFG)
Wissenschaftliche Ansprechpartner:
Prof. Dr. Wolfgang A. Linke
University of Münster
Institute of Physiology II
Linke Lab: Cardiovascular Physiology
Tel: +49 251 83-55328
wlinke@uni-muenster.de
Originalpublikation:
Christine M. Loescher, Martin Breitkreuz, Yong Li, Alexander Nickel, Andreas Unger, Alexander Dietl, Andreas Schmidt, Belal A. Mohamed, Sebastian Kötter, Joachim P. Schmitt, Marcus Krüger, Martina Krüger, Karl Toischer, Christoph Maack, Lars I. Leichert, Nazha Hamdani, Wolfgang A. Linke (2020): Regulation of titin-based cardiac stiffness by unfolded domain oxidation (UnDOx). Proc Natl Acad Sci USA. Doi: 10.1073/pnas.2004900117
Weitere Informationen:
https://www.medizin.uni-muenster.de/physiologieii/das-institut.html Institute of Physiology II at Münster University
Media Contact
All latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















