Highly prevalent gene variants in minority populations cause kidney disease
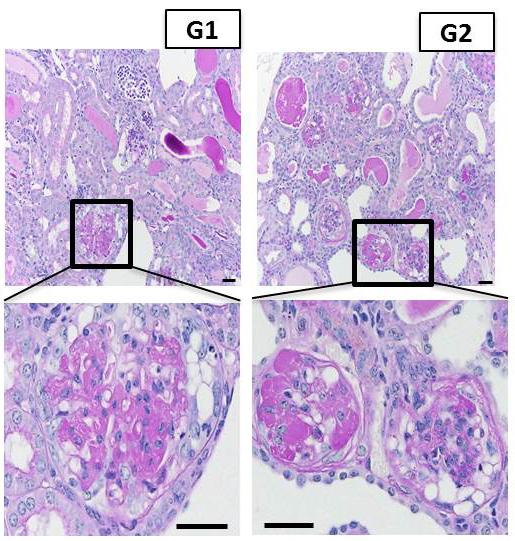
Representative images of kidney tissue from APOL1-G1 and APOL1-G2 mice, showing severe scarring of the kidney filter. Credit: Katalin Susztak, Perelman School of Medicine, University of Pennsylvania
African Americans have a heightened risk of developing chronic and end-stage kidney disease. This association has been attributed to two common genetic variants – named G1 and G2 — in APOL1, a gene that codes for a human-specific protein. However, direct evidence showing that these variants definitively cause kidney disease was lacking because APOL1 is widely expressed in different cell types but the gene is present in only some primates and humans. The challenge has been to create an animal model to prove this. Now, a team led by researchers from the Perelman School of Medicine at the University of Pennsylvania has engineered mice with these mutations that cause human-like kidney disease.
“The key missing piece has been whether these variants are true disease culprits,” said senior author Katalin Susztak, MD, PhD, an associate professor of Medicine and Genetics, of the study published online in Nature Medicine. “Our study established that these mutations are definitely disease causing.”
The G1 and G2 APOL1 gene variants, found almost exclusively in people of West African descent, have been shown to be associated with two-to-100-fold increased risk of kidney disease development, according to previous studies. Despite this highly significant risk, more than one third of African Americans carry the G1 and G2 variants. Biologists surmise that the reason these two mutations are so prevalent is that they emerged as a result of “positive selection” in people of African descent because the mutant proteins protect humans against the parasite that causes African sleeping sickness. Cells that express the G1 and G2 variants of the APOL1 protein are better able to kill these parasites.
To prove that expression of APOL1 with the G1 and G2 mutations causes kidney disease, the team made mice in which they could induce the expression of the non-mutated APOL1 gene as well as the G1 or G2 mutated APOL1 genes in different cell types. The team found that when the G1 and G2 variants are expressed in the filtering cells of the kidney the disease in the mouse model strongly resembled features of human kidney disease at the functional, structural, and molecular level. “These mutant proteins caused the kidney filter to become leaky and scarred, resulting in defective kidney function” Susztak said.
Kidney disease development was specific to the filtering cells of the kidney. The scientists found that G1 or G2 mutated APOL1 proteins interfere with the normal house-cleaning function of the cell, leading to an accumulation of jumbled proteins, inflammation, and eventually cell death. This trash removal system is especially important in kidney filtering cells, as these cells do not renew and losing them results in scarring of kidney tissue.
“Now that we know that the G1 and G2 mutated APOL1 proteins cause human-like kidney disease, we can start to look for ways to target them to reduce kidney disease risk among millions of people of African descent,” Susztak said. “The good news is that in mice the disease development was experimentally reversible when the G1 and G2 genes were turned off, and in a related finding, disease severity also correlated with the amount of expression of G1 and G2 APOL1 variant proteins in patient samples.”
###
Coauthors are Pazit Beckerman, Jing Bi-Karchin, Ae Seo Deok Park, Chengxiang Qiu, Patrick D. Dummer, Irfana Soomro, Carine M. Boustany-Kari, Steven S. Pullen, Jeffrey H. Miner, Chien-An A. Hu, Tibor Rohacs, Kazunori Inoue, Shuta Ishibe, Moin A. Saleem, Matthew B. Palmer, Ana Maria Cuervo, and Jeffrey B. Kopp.
This work was supported by the National Institutes of Health (DK105821, T32-DK007006).
Penn Medicine is one of the world's leading academic medical centers, dedicated to the related missions of medical education, biomedical research, and excellence in patient care. Penn Medicine consists of the Raymond and Ruth Perelman School of Medicine at the University of Pennsylvania (founded in 1765 as the nation's first medical school) and the University of Pennsylvania Health System, which together form a $5.3 billion enterprise.
The Perelman School of Medicine has been ranked among the top five medical schools in the United States for the past 18 years, according to U.S. News & World Report's survey of research-oriented medical schools. The School is consistently among the nation's top recipients of funding from the National Institutes of Health, with $373 million awarded in the 2015 fiscal year.
The University of Pennsylvania Health System's patient care facilities include: The Hospital of the University of Pennsylvania and Penn Presbyterian Medical Center — which are recognized as one of the nation's top “Honor Roll” hospitals by U.S. News & World Report — Chester County Hospital; Lancaster General Health; Penn Wissahickon Hospice; and Pennsylvania Hospital — the nation's first hospital, founded in 1751. Additional affiliated inpatient care facilities and services throughout the Philadelphia region include Chestnut Hill Hospital and Good Shepherd Penn Partners, a partnership between Good Shepherd Rehabilitation Network and Penn Medicine.
Penn Medicine is committed to improving lives and health through a variety of community-based programs and activities.
In fiscal year 2015, Penn Medicine provided $253.3 million to benefit our community.
Media Contact
All latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















