Dismantling pancreas cancer’s armor
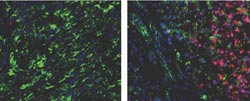
Red cancer-fighting T cells are rare in tumors rich in immunosuppressive cells (left), but rapidly enter tumors when immunosuppressive cells are removed (right).<br><br>Image credit: Hingorani Lab, Fred Hutchinson Cancer Research Center<br>
Pancreas cancer is notoriously impervious to treatment and resists both chemotherapy and radiotherapy. It has also been thought to provide few targets for immune cells, allowing tumors to grow unchecked.
But new research from Fred Hutchinson Cancer Research Center shows that pancreas cancer “veils” itself from the immune system by recruiting specialized immune suppressor cells. The research team also found that removing these cells quickly triggers a spontaneous anti-tumor immune response.
The findings, published Feb. 20, 2014 in Gut, give hope for future immunotherapy strategies against this deadly and aggressive cancer.
“The take-home message is that there is a latent immune response against pancreas cancer that can be expressed if we remove its obstacles,” said Sunil Hingorani, M.D., Ph.D., an associate member of the Clinical Research Division at Fred Hutch, who led the study. “Removing the suppressor cells creates a context that could enable an adoptive immune cell therapy against pancreas cancer.”
Activating a T-cell response against the cancer
Pancreas cancer is nearly always diagnosed at very late stages, which has made its development hard to study. To gain insight into these aggressive tumors, Hingorani’s team pioneered the development of a genetic mouse model of pancreas cancer. Previous work in the model led to their discovery of an enzyme that can make pancreas tumors more permeable to chemotherapy. The group turned again to this model to learn more about how pancreas tumors interact with the immune system.
From their earlier work, Hingorani’s team knew that several different types of immunosuppressive cells infiltrate pancreas tumors. Together with immunologist Philip Greenberg, M.D., a member of Fred Hutch’s Clinical Research Division, they have begun studying ways to target these inhibitory cells. As Ingunn Stromnes, Ph.D., the postdoctoral researcher co-mentored by Hingorani and Greenberg who spearheaded this latest study, watched pancreas tumors develop in mice, she saw that one cell type stood out. Descended from bone marrow cells and dubbed granulocyte-myeloid-derived suppressor cells (Gr-MDSCs), these cells jumped in number as pancreas tumors turned invasive. Stromnes discovered the pancreas tumors were orchestrating the accumulation of these suppressor cells by releasing a protein known as granulocyte macrophage colony-stimulating factor (GM-CSF), which attracted the Gr-MDSCs.
Strikingly, the Gr-MDSCs actively worked against T cells, a class of immune cell central to many immunotherapy strategies. T cells are often harnessed to fight tumors because they can recognize very specific molecules and destroy any cells expressing those molecules. But Gr-MDSCs prevented T cells from dividing and even induced their death.
Stromnes found this effect could be reversed, however, and the T-cell response activated, by depleting Gr-MDSCs. When she did so, she saw evidence not only that the T cells could now enter the tumors, but also that the tumors showed evidence of the type of cellular damage the T cells are designed to mete out.
“The findings are important because they show that the tumor microenvironment itself, and in particular a specific subset of cells in the tumor, is preventing T cells from trafficking to the tumor and mounting a response,” Stromnes said. Importantly, humans also possess cells very similar to Gr-MDSCs, which strengthens the case that similar strategies could impact human pancreas cancer. Additionally, the damage wreaked on Gr-MDSC-depleted tumors appeared to release some of the pressure inside the tumor, allowing crushed blood vessels to open again and providing a potential avenue for chemotherapy.
‘We want to put as big a hurt on pancreas cancer as possible’
The results are a backbone on which the team can begin designing a multipronged approach to pancreas cancer therapies, Stromnes noted. The findings show that a T-cell-based therapy alone may not be enough. Researchers must also take into account pancreas cancer’s immunosuppressive strategies. “We’re trying to get the helpful immune cells into the tumors, and our results show that to do that, we need to get rid of these inhibitory cells the tumors have co-opted,” she said.
The team is now working to develop a T-cell therapy to take advantage of their new findings. They plan to test their Gr-MDSC strategy combined with immunotherapy as well as chemotherapy to devise the strongest possible treatment for pancreas cancer.
“Our goal is not incremental advances,” Hingorani said. “We want to put as big a hurt on pancreas cancer as possible.”
Solid tumors, such as those of the pancreas, are the focus of Solid Tumor Translational Research, a network comprised of Fred Hutchinson Cancer Research Center, UW Medicine and Seattle Cancer Care Alliance. STTR is bridging laboratory sciences and patient care to provide the most precise treatment options for patients with solid tumor cancers. Collectively, these leading cancer research and clinical care organizations are enhancing the knowledge of cancer using population research, preclinical cancer biology, translational, imaging and clinical studies of solid tumors.
Work for the new study was supported by the National Cancer Institute, the Giles W. and Elise G. Mead Foundation, the Safeway Foundation, the Irvington Institute Fellowship Program of the Cancer Research Institute, the Jack & Sylvia Paul Fund to Support Collaborative Immunotherapy Research and the Fred Hutchinson/University of Washington Cancer Consortium.
Note for media: Images of immunosuppressive-cell-rich and cell-depleted pancreas tumors are available upon request. To obtain a copy of the Gut paper, “Targeted depletion of an MDSC subset unmasks pancreatic ductal adenocarcinoma to adaptive immunity,” or to arrange an interview with one of the authors, please contact Kristen Woodward at kwoodwar@fhcrc.org or 206-667-2210. (Citation: Stromnes, I.M. et al., Gut Published Online First: Feb. 20, 2014, doi: 10.1136/gutjnl-2013-306271.)
At Fred Hutchinson Cancer Research Center, home to three Nobel laureates, interdisciplinary teams of world-renowned scientists seek new and innovative ways to prevent, diagnose and treat cancer, HIV/AIDS and other life-threatening diseases. Fred Hutch’s pioneering work in bone marrow transplantation led to the development of immunotherapy, which harnesses the power of the immune system to treat cancer with minimal side effects. An independent, nonprofit research institute based in Seattle, Fred Hutch houses the nation’s first and largest cancer prevention research program, as well as the clinical coordinating center of the Women’s Health Initiative and the international headquarters of the HIV Vaccine Trials Network. Private contributions are essential for enabling Fred Hutch scientists to explore novel research opportunities that lead to important medical breakthroughs. For more information visit www.fredhutch.org or follow Fred Hutch on Facebook, Twitter or YouTube.
CONTACT
Kristen Woodward
206-667-2210
media@fredhutch.org
Media Contact
More Information:
http://www.fredhutch.orgAll latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















