Near-infrared imaging system shows promise as future pancreatic cancer diagnostic tool

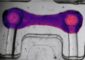
To test the diagnostic potential of OCT imaging, researchers used the technique to examine surgically removed pancreatic tissue samples from patients with cystic lesions. By identifying unique features of the high-risk cysts that appeared in the OCT scans, the team developed a set of visual criteria to differentiate between high and low risk cysts.
They then tested the criteria by comparing OCT diagnoses to those obtained by examining thin slices of the pancreatic tissue under a microscope. Their results, described in the August issue of the Optical Society's (OSA) open-access journal Biomedical Optics Express, showed that OCT allowed clinicians to reliably differentiate between low-risk and high-risk cysts with a success rate close to that achieved by microscope-assisted examinations of slices of the same samples.
Future studies by the research team will focus on improving imaging resolution to further differentiate between solid lesions and autoimmune pancreatitis, and test this technology in vivo. They recently received FDA approval for testing this technology in human patients by using an OCT probe small enough to be inserted into the pancreas through a biopsy needle, which will be guided into suspect masses in the pancreas by endoscopic ultrasound imaging. A pilot clinical study is planned to start within the next couple of months. If in vivo data will prove reliable differentiation between the two types of cysts, a study in a larger number of patients will be planned, contingent on NIH funding and FDA approval.
Paper: “Differentiation of pancreatic cysts with optical coherence tomography (OCT) imaging: an ex-vivo pilot study,” Biomedical Optics Express, Iftimia et al., Vol. 2, Issue 8, pp. 2372-2382. http://www.opticsinfobase.org/boe/abstract.cfm?URI=boe-2-8-2372
EDITOR'S NOTE: This summary is part of OSA's monthly Biomedical Optics Express tip sheet. To subscribe, email astark@osa.org or follow @OpticalSociety on Twitter. For images or interviews with authors, please contact Angela Stark, astark@osa.org or 202.416.1443.
About Biomedical Optics Express
Biomedical Optics Express is OSA's principal outlet for serving the biomedical optics community with rapid, open-access, peer-reviewed papers related to optics, photonics and imaging in the life sciences. The journal scope encompasses theoretical modeling and simulations, technology development, and biomedical studies and clinical applications. It is published by the Optical Society and edited by Joseph A. Izatt of Duke University. Biomedical Optics Express is an open-access journal and is available at no cost to readers online at http://www.OpticsInfoBase.org/BOE.
About OSA
Uniting more than 106,000 professionals from 134 countries, the Optical Society (OSA) brings together the global optics community through its programs and initiatives. Since 1916 OSA has worked to advance the common interests of the field, providing educational resources to the scientists, engineers and business leaders who work in the field by promoting the science of light and the advanced technologies made possible by optics and photonics. OSA publications, events, technical groups and programs foster optics knowledge and scientific collaboration among all those with an interest in optics and photonics.
Media Contact
All latest news from the category: Medical Engineering
The development of medical equipment, products and technical procedures is characterized by high research and development costs in a variety of fields related to the study of human medicine.
innovations-report provides informative and stimulating reports and articles on topics ranging from imaging processes, cell and tissue techniques, optical techniques, implants, orthopedic aids, clinical and medical office equipment, dialysis systems and x-ray/radiation monitoring devices to endoscopy, ultrasound, surgical techniques, and dental materials.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…