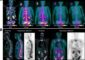
Automated analysis of MR images may identify early Alzheimer’s disease

In their report that will appear in the journal Brain and has been released online, the MGH/Martinos team show how their software program can accurately differentiate patients with mild cognitive impairment or Alzheimer's disease from normal elderly individuals based on anatomic differences in brain structures known to be affected by the disease.
“Traditionally Alzheimer's has been diagnosed based on a combination of factors – such as a neurologic exam, detailed medical history and written tests of cognitive functioning – with neuroimaging used primarily to rule out other diseases such as stroke or a brain tumor,” says Rahul Desikan MD, PhD, of the Martinos Center and Boston University School of Medicine, lead author of the Brain paper. “Our findings show the feasibility and importance of using automated, MRI-based neuroanatomic measures as a diagnostic marker for Alzheimer's disease.”
The researchers note that mild cognitive impairment occurs in about 20 percent of elderly individuals – as many as 40 percent of those over 85 – 80 percent of whom develop Alzheimer's within five or six years. Since drugs that may slow the progression of Alzheimer's are in development, the ability to treat patients in the earliest stages of the disease may significantly delay progression to dementia. To investigate whether MR imaging can produce diagnostic markers for mild cognitive impairment and Alzheimer's disease, the research team used FreeSurfer – an openly available imaging software package developed at the Martinos Center and the University of California at San Diego – to examine a number of neuroanatomic regions across a range of normal individuals and patients with mild cognitive impairment and Alzheimer's disease.
In the first phase of the study, the investigators examined MR images of 97 elderly individuals, some who had been determined to have mild cognitive impairment and others who were cognitively normal. Analyzing those images identified three regions of the brain where structural differences distinguished the normal controls from participants with mild cognitive impairment with an accuracy of 91 percent. Earlier pathological and imaging studies have found evidence of early Alzheimer's disease in these three areas – the hippocampus, entorhinal cortex and the supramarginal gyrus.
To validate the accuracy and assess the reliability of the first-phase observations, the investigators analyzed imaging data from 216 individuals in the Alzheimer's Disease Neuroimaging Database – 94 of whom were normal, 58 who had mild cognitive impairment at the time of imaging and went on to develop dementia, and 65 who had probable Alzheimer's based on their clinical symptoms. These participants also had a series of neuropsychological tests, and samples of cerebrospinal fluid were available for many of them.
Automated MRI measures of the same three areas identified in the first phase – entorhinal cortex, hippocampus, and supramarginal gyrus – discriminated individuals with mild cognitive impairment from normal elderly controls with 95 percent accuracy, and patients with Alzheimer's were discriminated from normal controls with 100 percent accuracy. The MRI measures also were significantly correlated with clinical and cognitive tests of dementia, particularly memory decline, and with biomarkers of cellular pathology such as the Alzheimer's-associated forms of the tau and amyloid proteins.
“Our results indicate that these automated MRI measures are one effective way of identifying individuals in the earliest stages of Alzheimer's disease, but before this technology can be used clinically, several follow-up studies need to be done,” says Desikan. “Those include determining whether these automated MRI measures can accurately predict which individuals with mild cognitive impairment will progress to Alzheimer's; seeing if they can differentiate Alzheimer's from other neurodegenerative diseases; assessing how these measures do at early diagnosis, compared to other measures such as cellular biomarkers; and then validating all of these findings against the gold standard for diagnosis, postmortem examination of brain tissue.”
Bruce Fischl, PhD, of the Martinos Center is the senior author of the Brain study. Additional co-authors are Nicholas Schmansky, Douglas Greve and David Salat of the Martinos Center; Howard Cabral, Boston University School of Public Health; Christopher Hess, William Dillon, Christine Glastonbury and Michael Weiner, University of California, San Francisco; and Randy Buckner, Harvard University and Howard Hughes Medical Institute. Support for the study came from the American Federation for Aging Research, the National Center for Research Resources, the National Institute for Biomedical Imaging and Bioengineering, the Mental Illness and Neuroscience Discovery Institute, the National Institute on Aging, the National Institute for Neurologic Disorders and Stroke and several other organizations.
Massachusetts General Hospital, established in 1811, is the original and largest teaching hospital of Harvard Medical School. The MGH conducts the largest hospital-based research program in the United States, with an annual research budget of more than $500 million and major research centers in AIDS, cardiovascular research, cancer, computational and integrative biology, cutaneous biology, human genetics, medical imaging, neurodegenerative disorders, regenerative medicine, systems biology, transplantation biology and photomedicine.
Media Contact
More Information:
http://www.mgh.harvard.eduAll latest news from the category: Medical Engineering
The development of medical equipment, products and technical procedures is characterized by high research and development costs in a variety of fields related to the study of human medicine.
innovations-report provides informative and stimulating reports and articles on topics ranging from imaging processes, cell and tissue techniques, optical techniques, implants, orthopedic aids, clinical and medical office equipment, dialysis systems and x-ray/radiation monitoring devices to endoscopy, ultrasound, surgical techniques, and dental materials.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…