UT Arlington research may unlock enzyme’s role in disease
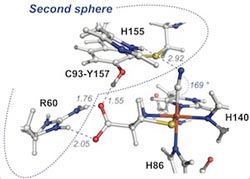
First and Second coordination spheres of the CDO active site.<br>
Brad Pierce, an assistant professor of chemistry/biochemistry at The University of Texas at Arlington, recently led a team that examined an oxygen utilizing iron enzyme called cysteine dioxygenase or CDO, which is found in high levels within heart, liver, and brain tissues.
Enzymes are proteins that act as catalysts to enable metabolic functions, but under some circumstances these oxygen-dependent enzymes can also produce highly toxic side products called reactive oxygen species or ROS.
For the first time, Pierce’s team found that mutations outside the CDO active site environment or “outer coordination sphere” have a profound influence on the release of ROS. Excess ROS has been linked to numerous age-onset human disease states.
“Most research in the past has focused on the active site inner coordination sphere of these enzymes, where the metal molecule is located,” said Pierce. “What we’re finding is that it’s really the second sphere that regulates the efficiency of the enzyme. In essence, these interactions hold everything together during catalysis. When this process breaks down, the enzyme ends up spitting out high levels of ROS and increasing the likelihood of disease.”
The study was published in December by the American Chemical Society journal Biochemistry. Pierce is corresponding author on the paper, with UT Arlington students Wei Li, Michael D. Pecore and Joshua K. Crowell as co-authors. Co-author Elizabeth J. Blaesi is a graduate research assistant at the University of Wisconsin.
Pierce believes the findings from the CDO enzyme could be applied to other oxygen-dependent enzymes, which make up about 20 percent of the enzymes in the human body.
“In principle, these findings could be extended to better understand how other enzymes within the class generate ROS and potentially be used to screen for genetic dispositions for ROS-related diseases,” he said.
Pierce’s research brings a new level of detail to enzyme study through the use of electron paramagnetic resonance or EPR, a technology similar to the magnetic resonance imaging or MRI used in the medical field. In fall 2012, the National Science Foundation awarded Pierce a three-year, $300,000 grant to study enzymes that are catalysts for the oxidation of sulfur-bearing molecules in the body.
“Dr. Pierce’s research is a good example of how basic science can set a path toward discoveries that affect human health. We look forward to his continued exploration of these findings,” said Pamela Jansma, dean of the UT Arlington College of Science.
The title of the Biochemistry paper is “Second-Sphere Interactions between the C93-Y157 Cross-Link and the Substrate-Bound Fe Site Influence the O2 Coupling Efficiency in Mouse Cysteine Dioxygenase.” It is available online here: http://www.ncbi.nlm.nih.gov/pubmed/24279989.
The University of Texas at Arlington is a comprehensive research institution of more than 33,300 students and 2,300 faculty members in the epicenter of North Texas. It is the second largest institution in The University of Texas System. Total research expenditures reached almost $78 million last year. Visit www.uta.edu to learn more.
The University of Texas at Arlington is an Equal Opportunity and Affirmative Action employer.
Media Contact: Traci Peterson, Office:817-272-9208,
Cell:817-521-5494, tpeterso@uta.edu
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















