Spheres can form squares
<br>
In an article just published on-line in PNAS, researchers from Wageningen University (Netherlands) report an exception to this rule: when small, micrometer-sized particles are placed on a curved oil-water surface, they arrange in a square pattern, as on a chessboard.
Since a number of decades, scientists are looking for strategies to create ordered crystal structures of regularly arranged small particles. Such crystals are interesting, because they can be used to control, modulate, or steer visible light in applications like lasers or other optical devices. While creating hexagonal patterns is relatively easy – this is the natural way in which the particles tend to order – anything different from that is much more difficult.
The team from the Laboratory of Physical Chemistry and Colloid Science at Wageningen University, part of Wageningen UR, has now found a way to create square particle arrays. To do this, they make use of the surface tension of the underlying oil-water surface, that is the tendency of the surface to minimize its area. When a particle sticks to the liquid surface it deforms the surface somewhat, and thereby increases the total area.
The surface tension acts to minimize these deformations, which can be done by clustering all the particles together. This effect is also responsible for the clumping of breakfast cereals in a bowl of milk or of bubbles at the surface of a soft drink. The researchers have found that this effective attraction between particles resulting from the surface tension depends on how the liquid surface is curved.
A slight curving of the surface already makes the interaction between particles highly dependent on their relative orientation, with attraction in two perpendicular directions and repulsion in the other directions. This is what causes the particles to arrange spontaneously in square patterns.
The researchers believe that their findings will lead to new bottom-up strategies for the design of structured materials, to be used in high-tech optical applications.
Publication
Capillarity-induced ordering of spherical colloids on an interface with anisotropic curvature.Dmitry Ershov, Joris Sprakel, Jeroen Appel, Martien A. Cohen Stuart, and Jasper van der Gucht. PNAS (early edition, May 2013). The research has been supported financially by the Netherlands Organisation for Scientific Research, NWO.
NOTE FOR THE EDITOR
For more information please contact prof.dr Jasper van der Gucht, Head of the Laboratory of Physical Chemistry and Colloid Science, tel. + 31 317 483066, jasper.vandergucht@wur.nl or Jac Niessen, science information officer, Wageningen UR, tel. + 31 317 485003, jac.niessen@wur.nl.
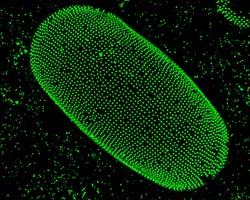
Caption: Particle organization on oil-water interface of a droplet. The particles are labeled with a fluorescent marker to make them visible.
Wageningen University is part of Wageningen UR (University & Research centre).The mission of Wageningen UR is ‘To explore the potential of nature to improve the quality of life’. Within Wageningen UR, nine research institutes – both specialised and applied – have joined forces with Wageningen University to help answer the most important questions in the domain of healthy food and living environment. With approximately 30 locations (in the Netherlands, Brazil, Chile, Ethiopia and China), 6,000 members of staff and 9,000 students, Wageningen UR is one of the leading organisations in its domain worldwide. The integral approach to problems and the cooperation between the exact sciences and the technological and social disciplines are at the heart of the Wageningen Approach.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Machine learning algorithm reveals long-theorized glass phase in crystal
Scientists have found evidence of an elusive, glassy phase of matter that emerges when a crystal’s perfect internal pattern is disrupted. X-ray technology and machine learning converge to shed light…

Mapping plant functional diversity from space
HKU ecologists revolutionize ecosystem monitoring with novel field-satellite integration. An international team of researchers, led by Professor Jin WU from the School of Biological Sciences at The University of Hong…

Inverters with constant full load capability
…enable an increase in the performance of electric drives. Overheating components significantly limit the performance of drivetrains in electric vehicles. Inverters in particular are subject to a high thermal load,…





















