Silenced – Overcoming multidrug resistance in cancer cells by silencing genes with RNA
<br>
In the journal Angewandte Chemie, a team of Chinese and American researchers has now introduced a new approach to gene therapy that could counter this problem: The gene that codes for resistance is “silenced” through the use of an ingenious nanocomplex.
Every cell in our body contains our complete genetic information. However, not all genes are used in every cell at all times. Regulatory processes are needed to determine when a gene should be read and transcribed to messenger-RNA (mRNA), and the corresponding protein built.
One such mechanism is RNA silencing. In this mechanism, short, specific, silencing RNA (siRNA) fragments bind to the mRNA to be silenced with participation from several enzyme complexes. The enzymes cleave the mRNA, preventing its translation into a protein. Gene therapies based on synthetic siRNA are under clinical development.
However, these siRNA drugs are directed toward the cellular silencing “machinery” and may disrupt natural gene regulation pathways, which results in side effects. In addition, they require a transport system to carry them through the cell membrane and to protect them from rapid degradation. Led by Min Yang at the Jiangsu Institute of Nuclear Medicine (Wuxi, China) and Xiaoyuan Chen at the National Institutes of Health (Bethesda, USA), the researchers have now developed an alternative approach that doesn’t have these disadvantages. It is based on a nanocomplex that already includes the required machinery and packaging.
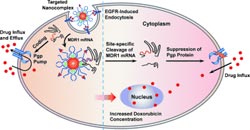
The researchers chose to use gold nanoparticles as their support and transport system. They attached three components to the nanoparticles’ surfaces: 1) RNAse A, a robust enzyme that nonspecifically cleaves single-stranded RNA; 2) DNA oligonucleotides with a sequence selected to specifically bind the mRNA to be taken out of circulation; 3) A ligand that is designed to pilot the nanocomplex to the target cells – tumor cells in this case. The scientists chose Cys-tag EGF, a ligand that binds to a growth-hormone receptor present in significantly elevated quantities in the cell membranes of many tumors.
One important mechanism of multidrug resistance in tumor cells is the active expulsion of drugs by means of a special transport protein (Pgp). Administration of chemotherapy drugs triggers formation of a large number of these transporters, which effectively protect the tumor cells from the drugs.
In order to silence the gene that codes Pgp, the researchers incorporated DNA that recognizes the corresponding mRNA into the nanocomplexes.
They were thus able to observe cleavage of this mRNA, a reduction in the concentration of Pgp, and renewed sensitivity toward the chemotherapy drug doxorubicin in multidrug-resistant tumor cell lines. In addition to combating multidrug resistance, the new method should prove to be a generally useful approach for gene therapy.
About the Author
Dr. Xiaoyuan (Shawn) Chen is Senior Investigator and Chief of the Laboratory of Molecular Imaging and Nanomedicine (LOMIN) at the National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health. His aims are to develop molecular imaging toolbox for better understanding of biology, early diagnosis of disease, monitoring therapy response, and guiding drug discovery/development. His lab puts special emphasis on high-sensitivity nanosensors for biomarker detection and theranostic nanomedicine for imaging, and gene and drug delivery.
Author: Xiaoyuan Chen, National Institutes of Health (NIH), Bethesda (USA), http://www.nibib.nih.gov/about-nibib/staff/xiaoyuan-shawn-chen
Title: Biomimetic RNA-Silencing Nanocomplexes: Overcoming Multidrug Resistance in Cancer Cells
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201309985
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…
Innovative microscopy demystifies metabolism of Alzheimer’s
Researchers at UC San Diego have deployed state-of-the art imaging techniques to discover the metabolism driving Alzheimer’s disease; results suggest new treatment strategies. Alzheimer’s disease causes significant problems with memory,…
A cause of immunodeficiency identified
After stroke and heart attack: Every year, between 250,000 and 300,000 people in Germany suffer from a stroke or heart attack. These patients suffer immune disturbances and are very frequently…





















