Sensitive balance in the immune system
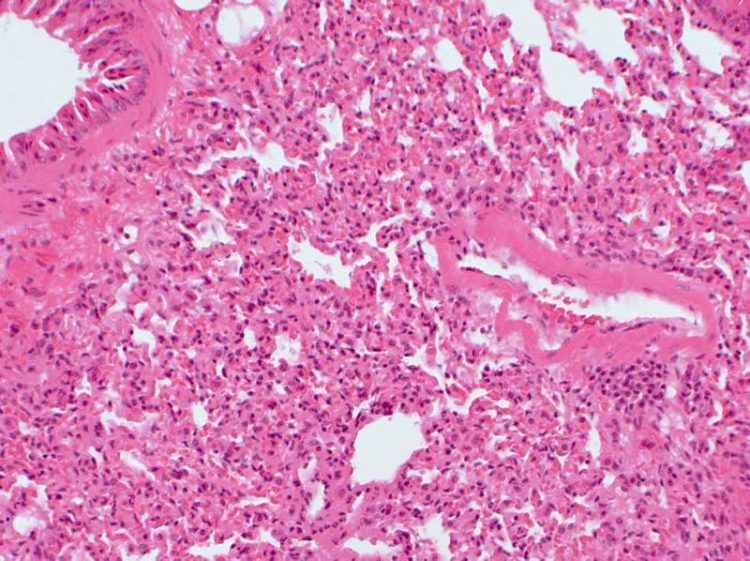
Inflammed lung tissue with immune cells that have migrated into the lungs. Their nuclei are stained blue. HZI/Pils
As strange as it may seem, a process called “cellular suicide” is actually crucial for the survival of the entire body. A protein called c-FLIPR plays a key role in controlling this process, which is called “apoptosis”.
Scientists at the Helmholtz Centre for Infection Research (HZI) have described the significance of c-FLIPR for the immune system in more detail: In the presence of an excess of this molecule, mice can fight infectious diseases better, but they develop autoimmune diseases as they get older. The inhibitory effect of c-FLIPR on apoptosis is the underlying cause of both these effects.
Apoptosis is used by cells that are changed by disease or are simply not needed any longer to eliminate themselves before they become a hazard to the body—on a cellular level, death is part of life. Disruption of this process can lead to cancer or immunodeficiencies, but also to autoimmune diseases, in which cells attack their own body.
HZI scientist Prof Ingo Schmitz and his team investigate the regulation of apoptosis in the immune system. In collaboration with researchers of the Otto von Guericke University Magdeburg and the Helmholtz Zentrum München, they elucidated the role of a central protein in this process.
The researchers published their results in “Cell Death & Disease”. So-called c-FLIP proteins inhibit signaling cascades that can lead to apoptosis. This is important temporarily in the response to pathogens to ensure that lymphocytes, a type of immune cells, can proliferate sufficiently. Towards the end of the immune response, once the lymphocytes completed their tasks and successfully eliminated the pathogen, c-FLIP is usually degraded. As a result, apoptosis is enabled again, the lymphocytes die and the equilibrium in the immune system is restored.
The HZI researchers then took a closer look at the exact function of a certain variant of the protein, called c-FLIPR. They used mice to investigate what happens if this protein is always present in lymphocytes and other blood cells. Whereas the apoptosis inhibitor caused no anomalies in young mice, the scenario in older mice was quite different: “The composition of the lymphocytes was changed significantly,” says Schmitz. “Furthermore the immune cells were strongly activated.”
The overactivation is easily apparent in the body. The researchers found immune molecules, called autoantibodies, which attack the body's own tissue in the kidneys and lung. In addition, they detected harmful protein deposits in the kidneys. The changes in the lung tissue are also indicative of the immune system attacking its own body in the presence of too much c-FLIPR. “Immune cells migrate into the lung and attack the lung tissue,” says Schmitz. Physicians usually see these symptoms in a human autoimmune disease called systemic lupus erythematosus
The HZI scientists discovered already last year that cells can fight bacterial infections better if c-FLIPR is turned on permanently. This means that inhibiting the suicide of cells has beneficial effects in acute infections, but leads to autoimmune reactions in the long run. “c-FLIPR is important for the balance of the immune system. It might be possible to intervene with suitable therapeutic agents if the equilibrium of the immune system is disrupted,” says Schmitz.
Original publication
Frida Ewald, Michaela Annemann, Marina C. Pils, Carlos Plaza-Sirvent, Frauke Neff, Christian Erck, Dirk Reinhold, Ingo Schmitz
Constitutive expression of murine c-FLIPR causes autoimmunity in aged mice
Cell Death & Disease, 2014
The research group “Systems-oriented Immunology and Inflammation Research” studies the molecular processes that sensitize immune cells to the body’s own tissues. A main focus is on the cellular suicide programme apoptosis.
The Helmholtz Centre for Infection Research (HZI)
Scientists at the Helmholtz Centre for Infection Research in Braunschweig, Germany, are engaged in the study of different mechanisms of infection and of the body’s response to infection. Helping to improve the scientific community’s understanding of a given bacterium’s or virus’ pathogenicity is key to developing effective new treatments and vaccines.
http://www.helmholtz-hzi.de
http://www.helmholtz-hzi.de/en/news_events/news/view/article/complete/sensitive_… – This press release on www.helmholtz-hzi.de
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Machine learning algorithm reveals long-theorized glass phase in crystal
Scientists have found evidence of an elusive, glassy phase of matter that emerges when a crystal’s perfect internal pattern is disrupted. X-ray technology and machine learning converge to shed light…

Mapping plant functional diversity from space
HKU ecologists revolutionize ecosystem monitoring with novel field-satellite integration. An international team of researchers, led by Professor Jin WU from the School of Biological Sciences at The University of Hong…

Inverters with constant full load capability
…enable an increase in the performance of electric drives. Overheating components significantly limit the performance of drivetrains in electric vehicles. Inverters in particular are subject to a high thermal load,…





















