How scavenging fungi became a plant's best friend
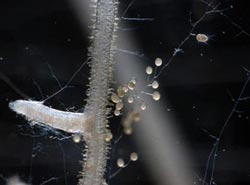
These are spores and hyphae (root-like extensions) of an arbuscular mycorrhizal fungi (AMF), R. irregularis, grown among carrot hairy roots.<br><br>Credit: Guillaume Bécard (University of Toulouse)<br>
More than two thirds of the world's plants depend on this soil-dwelling symbiotic fungus to survive, including critical agricultural crops such as wheat, cassava, and rice.
The analysis of the Rhizophagus irregularis genome has revealed that this asexual fungus doesn't shuffle its genes the way researchers expected. Moreover, rather than having lost much of its metabolic genes, as observed in many mutualistic organisms, it has expanded its range of cell-to-cell communication genes and phosphorus-capturing genes.
A team led by the French National Institute for Agricultural Research (INRA) and including researchers from the Department of Energy Joint Genome Institute (DOE JGI) reported the complete genome of R. irregularis (formerly Glomus intraradices) in a paper published online November 25 in the journal Proceedings of the National Academy of Sciences (PNAS).
The fungus is a member of the Glomeromycota family and frequently colonizes many plants important to agriculture and forestry. Glomeromycota, also called arbuscular mycorrhizal fungi (AMF), play a vital role in how phosphorus and carbon cycles through the atmosphere and land-based ecosystems, but exactly how it does this vital job is poorly understood.
“This is the first sequenced genome of arbuscular mycorrhizae, the type that is dominant on the planet,” said Igor Grigoriev, one of the senior authors on the paper and lead for the Fungal Genomics Program at the DOE JGI.
It was a long hard road to a sequenced arbuscular mycorrhizal fungus. In 2006, shortly after the DOE JGI sequenced the first tree genome, Populus trichocarpa, it became apparent that it took a village (of other organisms) to raise a poplar tree. Researchers Jerry Tuskan of Oak Ridge National Laboratory and Francis Martin of INRA, recommended that the assembly of Populus-associated fungi and bacteria be sequenced to inform research on perennial plant growth, ecosystem function and plant microbe interactions. This long passage is outlined in an earlier publication in New Phytologist. Rhizophagus irregularis, is the next in this linage to be released by the DOE JGI, it follows the ectomycorrhizal fungal symbiont Laccaria, the poplar rust pathogen Melampsora, and dozens of bacterial genomes.
A relic of fungal evolution, AMF diverged early on from other forms of fungus. They form dense clusters of branched structures — called arbuscules — in root cells, much like a tight, many-fingered handhold. The arbuscules are the main route of nutrient exchange between plants and fungi. Unable to live on their own, AMF are entirely dependent on their plant hosts for the sugars they need for food. They have carefully established their relationship with host plants, keeping them alive while sapping nutrients from them.
But AMF are also adept at capturing phosphorus from the soil and making it available for their hosts. Phosphorus, a critical element for cellular function, is otherwise difficult to extract from the soil and is often the limiting factor for how quickly a plant grows.
Scientists theorize that the benefits these fungi provided enabled ancient plants to evolve during the Paleozoic era, about 250 to 500 million years ago. Over time, plants adapted their essentially rootless primordial form and developed deeper and stronger roots to take advantage of the nutrients that underground AMF fed them. In exchange, plants provided nutrients the fungi couldn't obtain themselves.
Analysis of the R. irregularis genome also revealed several surprising details. The research team found that the genome is among the largest fungal genomes sequenced, weighing in at 153 million base pairs (Mb). For comparison, the button mushroom (Agaricus bisporus), also sequenced and published by the DOE JGI, has a genome of about 30 Mb. Through several generations, portions of R. irregularis's genome were duplicated, invaded by repeated transposable elements, famously known as 'jumping genes'. Unlike many other fungi, R. irregularis seems to lack mechanisms that can keep these transposable elements from running amok.
“Among the expanded portions of its genome, R. irregularis had several genes for phosphorus metabolism, which are probably responsible for its large appetite for phosphorus,” said Francis Martin, one of the senior authors on the paper and lead for the Cluster of Excellence, Advanced Research on the Biology of Tree and Forest Ecosystems (ARBRE) at the INRA. “They also have an abundance of genes for communication between cells via signaling proteins, including small secreted effectors highly expressed during symbiosis. Plant roots send out a plethora of chemical signals and these genes probably help AMF interact with plants, picking up the signals plants pump out.”
Another surprise for the research team was in the genes that govern metabolism. “Obligate parasites often have broken metabolism, missing some genes in critical metabolic pathway which make them dependent on their host,” Grigoriev said. “We did not find such genes here.” R. irregularis has retained much of its metabolic machinery, unlike many other obligate parasitic organisms. It leads a double-life, extracting minerals from the soil while still living in harmony with its host plant.
Though it has nearly 30,000 protein-encoding genes, R. irregularis has also lost hundreds of genes as a result of its close association with plants. For example, it can't make most of the toxins other plant-interacting fungi release, probably, the researchers speculate, to avoid setting off the host plant's immune system. It has also cast off most of its genes for breaking down plant cell walls, a critical ability for free-living fungi that feed off dead organic matter in soils.
Teasing apart the complex relationship between soil fungi and plants is likely to have an impact on improving biofuel production from plant biomass. “Through analysis of this and other mycorrhizal genomes, we can help to better understand interactions and conditions critical for a sustainable growth of bioenergy plants, but also staple crops, a prerequisite to help feeding the world,” said Martin.
Learn more from researchers Grigoriev, Martin and other collaborators on the importance of fungal genomics in this video: http://bit.ly/JGI-Fungal-vid
The U.S. Department of Energy Joint Genome Institute, supported by the DOE Office of Science, is committed to advancing genomics in support of DOE missions related to clean energy generation and environmental characterization and cleanup. DOE JGI, headquartered in Walnut Creek, Calif., provides integrated high-throughput sequencing and computational analysis that enable systems-based scientific approaches to these challenges. Follow @doe_jgi on Twitter.
DOE's Office of Science is the largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Media Contact
More Information:
http://www.lbl.govAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















