A step closer to understanding how calcium controls our bodies
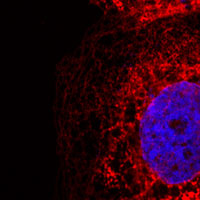
Cells expressing inositol triphosphate receptors with an engineered binding site stained with snake venom
Calcium signals control almost every activity in the human body, from fertilization to cell death and everything between, including every beat of the heart. The researchers have found that just 2-3 calcium channels, from among the many thousands present on the surface of a cell, are responsible for much of the calcium signal that regulates the activity of immune cells. The 2-3 channels are formed by inositol trisphosphate (IP3) receptors on the membrane that surrounds the cell. Despite being the cellular equivalent of a needle in a haystack, the team discovered that these 2-3 channels make a substantial contribution to calcium signalling.
The membranes of cells are like dams holding back a flood of calcium. Channels within these membranes are the sluice gates that cells regulate to allow controlled entry of calcium. Too much calcium and the cell dies. But just as the water from a dam may be directed via the sluice gates to a fish ladder or a generator, then so the calcium passing through channels can be sent to different intracellular proteins to bring about different cellular responses. In this way, the same intracellular messenger, calcium, can be used to control all sorts of things without confusing the cell. The new research shows that IP3 channels, originally thought to be found only in membranes within the cell, such as the endoplasmic reticulum, are also found in its external membrane.
The researchers believe that this new role for IP3 receptors on the cell membrane, additional to their established role within the cell, paves the way to addressing the mechanisms that allow calcium to so effectively regulate so many cellular activities. Such an understanding could give a better idea of processes governing cell growth and development and could eventually lead to more sophisticated drug targeting.
The scientists built on work from others that aimed to develop new treatments for venomous snake-bites, by making short peptides that specifically bind to the venom. Using molecular biological methods they incorporated the same peptide into the IP3 receptor and demonstrated that it was now regulated by the snake venom.
Professor Colin Taylor led the research at the University of Cambridge’s Department of Pharmacology. He said: “Every schoolchild knows that calcium is the element that forms our teeth and bones, but it also has this key and very dynamic role as a controlling signal within every cell of the human body. Everything from the beating of the heart to cell division and cell death is controlled by calcium. The key question is how cells manage to use calcium to control so much without getting confused. It’s rather like trying to listen in to just one conversation at a noisy party. Our research brings us another step forward to understanding how cells regulate the inward flow of calcium. That is important, because when we use calcium in this way as a signal, we invite the enemy within and unless we keep it under very tight control the calcium will kill the cell.
“One of the more intriguing features of the work is the reliability with which these cells incorporate such small numbers of IP3 receptors into the membrane. Even if we force the cells to produce more IP3 receptors, they still place just 2-3 of them in the plasma membrane, and they always seem to get it right. What we really don’t understand is how they do it. The challenge now is to find out.”
The research published in Science is based on work funded by BBSRC and the Wellcome Trust.
Media Contact
More Information:
http://www.bbsrc.ac.ukAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















