Natural tumor suppressor in body discovered by UCSD medical researchers
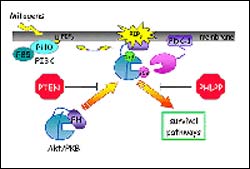
Akt/protein kinase B controls the balance between cell survival and cell death. The activated form of this kinase tips the balance towards cell proliferation and survival, whereas the inactive form tips the balance towards programmed cell death, apoptosis. Akt can be inactivated by two mechanisms: the tumor suppressor PTEN pre-empts activation by removing the activating signals, and the tumor suppressor PHLPP terminates activation by directly turning off a key phosphorylation switch on Akt.
A natural tumor suppressor that could potentially be turned on in certain cancer cells to prevent the formation of tumors has been discovered by researchers at the University of California, San Diego (UCSD) School of Medicine.
Located on chromosome 18 and called PH domain Leucine-rich repeat Protein Phosphatase (PHLPP, pronounced “flip”), the tumor suppressor is described in the April 1, 2005 issue of the journal Molecular Cell. The scientists demonstrated that PHLPP deletes a phosphate molecule, causing termination of cell-growth signaling by a protein called Akt that controls the balance between cell growth leading to cancer and cell death that prevents tumor formation.
A drug that turns on PHLPP, so that it suppresses cell growth caused by Akt, could be a potential cancer therapy,” said the study’s senior author, Alexandra C. Newton, Ph.D., UCSD professor of pharmacology. “Currently there are no compounds identified to directly stop Akt from causing cancer growth, once Akt signaling has been initiated.”
Scientists have known that Akt is critical in regulating cell growth and death, and that it is linked to some of the most common human cancers. Although one group of scientists discovered a molecule called PTEN*, which prevents activation of Akt, no one to date had determined how to directly turn off Akt once it has been activated.
Since the Akt molecule is locked in the “on” position when it has phosphate on it, the UCSD team reasoned that there must be another molecule that will strip off the phosphate and lock Akt in the “off” position. The scientists conducted a database search of the human genome for a phosphatase, which is an enzyme that acts as a catalyst in regulating cellular processes by removing phosphate molecules. Based on the chemical components of Akt, they specifically looked for a phosphatase linked to the PH domain, a protein module found in a wide variety of chemical signaling proteins in organisms ranging from yeast to humans.
Once they found PHLPP, which they discovered was expressed throughout the body, the scientists used biochemical and cellular studies in human and other mammalian tissue to determine that PHLPP levels are markedly reduced in several colon cancer and gliobastoma human cell lines that had elevated Akt phosphorylation. Reintroduction of PHLPP into the cell lines caused a dramatic suppression of tumor growth. With additional laboratory tests, the team found that PHLPP stops tumor growth by deleting a specific phosphate molecule at a position called Ser473 on Akt.
The scientists noted that PHLPP’s role as a tumor suppressor would apply to all cancers where Akt is elevated, “which is a large number of cancers,” Newton said.
Media Contact
More Information:
http://www.ucsd.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















