A Screening Strategy Using Zebrafish Targets Genes That Protect Against Hearing Loss

In a study published on February 29 in the open-access journal PLoS Genetics, researchers at the University of Washington have developed a research method that relies on a zebrafish’s lateral line—the faint line running down each side of a fish that enables it to sense its surroundings—to quickly screen for genes and chemical compounds that protect against hearing loss from some medications. The study was funded in part by the National Institute on Deafness and Other Communication Disorders (NIDCD), one of the National Institutes of Health.
“The fish’s lateral line contains sensory cells that are functionally similar to those found in the inner ear, except these are on the surface of the fish’s body, making them more easily accessible,” said James F. Battey, Jr., M.D., Ph.D., director of the NIDCD. “This means that scientists can very efficiently analyze the sensory structures under different conditions to find out what is likely to cause damage to these structures and, conversely, what can protect them from damage.”
When people are exposed to some antibiotics and chemotherapy agents, the sensory structures in the inner ear, called hair cells, can be irreversibly damaged, resulting in hearing loss and balance problems. These are known as ototoxic medications. People vary widely in their susceptibility to these agents as well as to damage caused by other chemical agents, loud sounds and aging.
To find out why this is so, senior scientists Edwin Rubel, David Raible, and their research team developed a screening strategy that uses hair cells in the lateral line of zebrafish larvae to signal how hair cells in a person’s inner ear might respond under similar conditions. Hair cells are named for small bristly extensions, or stereocilia, jutting from their tops. Movement of fluid (triggered by sound vibrations in the inner ear or changes in water pressure in the fish’s environment) causes the stereocilia to tilt to one side, generating an electrical impulse that travels to the brain.
The researchers first set out to identify genes that may be involved in how hair cells respond to ototoxic medicines. Using a chemical that causes random mutations in zebrafish, the researchers bred various fish families, with each family exhibiting a different set of mutations. The researchers then exposed five-day-old larval offspring to the drug neomycin, a type of antibiotic that damages these hair cells as well as those in the human inner ear. The larvae were then stained to determine if the hair cells were still intact. Fish that were resistant to damage were quickly identified as were those that were especially vulnerable.
Using genetic techniques, the group then examined the larvae’s DNA, searching for segments that were closely tied to the desired property. In doing so, they zoomed in on five mutations—each located on different genes—that, when inherited from each parent, protected against hair cell damage. Further examination revealed that one of the identified genes corresponds to a gene that is also found in other vertebrates, including humans. Another five mutations were identified that offer protection under more complex genetic conditions.
Next, the team investigated whether they could identify chemical compounds that protect hair cells against ototoxic medicines. Using the same screening technique—exposing five-day-old zebrafish larvae to neomycin and later applying special stains to the hair cells—the researchers screened more than 10,000 compounds and narrowed them down to two similar chemicals that provide robust protection of hair cells against the neomycin. One of the compounds was later found to protect hair cells from a mouse’s inner ear against the drug, indicating that the same compound may be protective for other mammals as well.
“One of the pluses about working with zebrafish is that, like other fish, they produce hundreds of offspring. We can look at lots of animals and we can look at many hair cells per animal, which means that we can get good quantitative data,” said Dr. Raible.
The authors suggest that their research technique, which combines chemical screening with traditional genetic approaches, offers a fast and efficient way to identify potential drugs and drug targets that may one day provide therapies for people with hearing loss and balance disorders.
Other sponsors of the study include the American Academy of Otolaryngology—Head and Neck Surgery Foundation, the University of Washington Royalty Research Fund and the V.M. Bloedel Hearing Research Center.
Media Contact
More Information:
http://www.plosgenetics.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Wildfire danger to increase due to climate change
WSL Institute for Snow and Avalanche Research (SLF) researchers expect an elevated wildfire danger in the Alpine Foreland from 2040 onwards due to changing meteorological conditions. The danger currently remains…
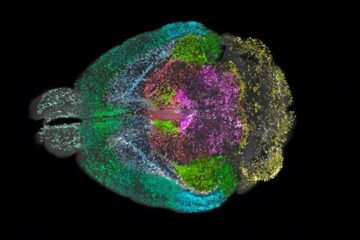
Advanced Brain Science Without Coding Expertise
Researchers at Helmholtz Munich and the LMU University Hospital Munich introduce DELiVR, offering a new AI-based approach to the complex task of brain cell mapping. The deep learning tool democratizes…
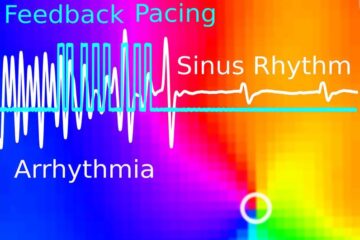
Gentle defibrillation for the heart
Using light pulses as a model for electrical defibrillation, Göttingen scientists developed a method to assess and modulate the heart function. The research team from the Max Planck Institute for…





















