Recent Reports Regarding Mercury and High Fructose

In summary we found:
The IATP report and Environmental Health article it references fall well below standards for proper scientific research and published literature.
The authors of both publications provide incomplete data and misleading conclusions.
Methods described by the authors deviate from standard procedure in testing for mercury.
The authors ignore important distinctions between organic and other forms of mercury and their implications for assessing human health risk.
Even if it were assumed that the mercury content found in the extremely limited sampling of foods and beverages was representative, the amounts are far lower than levels of concern set by government agencies.
The authors assume that the total mercury they detected in a questionably small sampling of consumer foods is primarily the result of high fructose corn syrup; an assumption that has not been properly tested or validated. The recipes for the items studied may have had multiple sources of potential contamination
To imply that there is a safety concern to consumers based on the findings presented is both incorrect and irresponsible.
By combining the results of a four-year-old sampling analysis of high fructose corn syrup with a more recent testing of branded foods and beverages for total mercury, the IATP report fails to recognize basic scientific facts regarding mercury; ignores common dietary sources of mercury, an element that is widely present in our environment at low concentrations; and makes improper assumptions regarding the source of the mercury measured in various branded food products.
More than two-thirds of the samples analyzed by IATP had no detectable level of mercury at all. In the remaining sample, most of these were at or near the limit of detection. The average concentration for the 17 samples with detectable levels was only 128 parts per trillion (ppt). EPA sets limits for mercury in drinking water at two parts per billion.
It is well known that small amounts of mercury are broadly present in our environment. For example, Health Canada reported in 2003 that the concentrations of total mercury in steak ranged from 420 to 1,800 parts per trillion (ppt); fresh pork contained 1,100 to 1,500 ppt; organ meats (liver and kidney) contained over 2,100 ppt; and lamb contained 290 to 2,300 ppt of total mercury. (Dabeka et al, 2003) For the sake of reference, one part per trillion is equal to one drop of water spread out into 26 Olympic-size swimming pools. (Washington Suburban Sanitary Commission, 2009)
That same study by Health Canada looked at mercury in seafood, finding amounts that ranged from 40,000 ppt in fresh or frozen marine fish to 148,000 ppt in canned fish. Other foods, such as canned mushrooms, had 5,100 to 16,000 ppt total mercury, grapes had 180 to 590 ppt, blueberries 210 to 640 ppt, rice 570 to 1,800 ppt, raisins upwards of 700 ppt, and shelled seeds up to 1,000 parts per trillion (ppt).
The levels noted above are substantially greater than those found by Wallinga and colleagues in their reports. (Dabeka et al, 2003) Other studies by other international authorities (FDA, United Kingdom Food Standards Agency and others) have found similar or higher levels of mercury in common components of a typical human diet. (FDA, 2006; Ysart et al, 2000)
IATP assumes that the total mercury they detected in a questionably small sampling of consumer foods is primarily the result of high fructose corn syrup; an assumption that has not been properly tested or validated. In fact, the authors do not attempt to characterize whether there may be mercury in any other ingredients contained within the consumer products tested, even while the recipes for the items studied may have had multiple sources of potential contamination. As we have mentioned, very small amounts of mercury are practically ubiquitous in our society.
IATP demonstrates a gross lack of understanding of the current state of knowledge of the different forms of mercury and their effect on the human body. his is illustrated in their statement that “The mercury found in HFCS may be a different form of mercury than the methylmercury typically found in fish (we just don’t know), but it poses a risk just the same. Mercury in any form can be toxic to the developing brain.” (Wallinga et al, 2009).
The ATSDR (Agency for Toxic Substances and Disease Registry) is a sister agency of the Centers for Disease Control, and regularly provides reviews of chemicals in our society that may pose a risk to human health. Regarding mercury, it is clear that inorganic forms are considered to be much less dangerous to humans than organic forms, with absorption generally at least ten times less (ATSDR, Public Health Statement on Mercury, 1999).
Dr. Dennis Paustenbach is a board-certified toxicologist and industrial hygienist with nearly 25 years of experience in risk assessment, environmental engineering, ecotoxicology, and occupational health. He is currently the President of ChemRisk, Inc., a consulting firm which specializes in human and ecological risk assessment and risk analysis of pharmaceuticals and medical devices. ChemRisk’s professionals have a longstanding reputation for thorough scientific analysis and for sharing their results both at major scientific meetings and in the peer-reviewed scientific literature. ChemRisk’s more than 60 scientists are experienced in addressing health and safety concerns, with backgrounds including toxicology, industrial hygiene, epidemiology, ecotoxicology, environmental sciences, medicine, statistical analysis, and risk assessment. Many of the more than 1,000 papers presented at scientific conferences and 400 papers published by ChemRisk® scientists are frequently referenced in regulatory decision-making and relied upon in litigation proceedings.
Media Contact
More Information:
http://www.chemrisk.comAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…
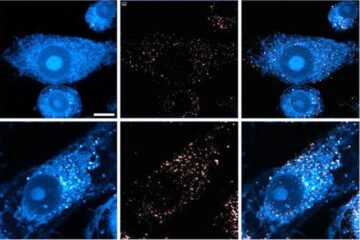
Innovative microscopy demystifies metabolism of Alzheimer’s
Researchers at UC San Diego have deployed state-of-the art imaging techniques to discover the metabolism driving Alzheimer’s disease; results suggest new treatment strategies. Alzheimer’s disease causes significant problems with memory,…
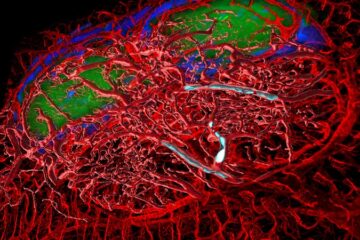
A cause of immunodeficiency identified
After stroke and heart attack: Every year, between 250,000 and 300,000 people in Germany suffer from a stroke or heart attack. These patients suffer immune disturbances and are very frequently…





















