"Prehistoric relics" as targets for vaccination against cancer
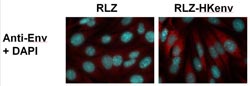
The murine renal carcinoma cell line Renca. On the left: without HERV-K envelope protein expression. On the right: genetically modified Renca cells with HERV-K envelope protein (in red).<br><br><br>Source: PEI - The image is available with 300 dpi (4,7x13,7 cm) - please ask the PEI Press and information office<br>
With a vaccine directed against endogenous retroviruses, the progression of tumour growth could be significantly inhibited in a mouse model. The prophylactic vaccination could even prevent the development of tumours. The results of this research project were reported in the online edition of PLoS ONE dated 30 August 2013 (11 p.m. CET).
Human endogenous retroviruses (HERV) entered the human genome millions of years ago and have been passed on from generation to generation. The human genome contains several thousand such endogenous retrovirus genes. As a rule, the genes of HERV are mute, i.e. they do not produce proteins. Researchers of the Paul-Ehrlich-Institut and other groups have already been able to show that an increase in expression of genes of one group of these retroviruses occurs in HIV infected individuals but also in the tumour cells of various types of cancer. The HERV group could be identified as HERV-K/HML-2(hom), or for short, HERV-K. Professor Dr Barbara Schnierle and her co-workers in the Virology Division of the Paul-Ehrlich-Institut now wanted to know whether the specific gene activity of HERV-K could be used therapeutically to fight cancer cells in a targeted manner. To protect healthy cells, it is required to identify structures as targets which are only displayed on cancer cells.
For their investigations, Professor Barbara Schnierle, head of the Section “AIDS, New and Emerging Pathogens”, and her co-workers used a murine kidney cell line (Renca). This cell line had been genetically modified in such a way that the cells produced (human) HERV-K envelope proteins to emulate the situation of the HERV-positive human cancer cells as accurately as possible. Mice which received an i.v. administration of these cells developed lung metastases within a short period of time.
For the “vaccination” against these tumours, the researchers used the modified vaccinia virus Ankara (MVA), an attenuated virus which cannot replicate after inoculation. In addition, Professor Schnierle and her co-workers had inserted the gene for the HERV-K envelope protein into the MVA genome. After the inoculation, the gene is read in the cells and the envelope protein is formed. It is presented to the immune system as antigen and an immune response can be induced.
Then the PEI scientists performed investigations to establish whether their “vaccine” could be used both for therapeutic and for prophylactic purposes: For the therapeutic approach, they first injected the genetically modified mouse cancer cells, then waited ten days until lung metastases were formed and then treated part of the mice with the MVA vaccine. The tumours grew considerably more slowly in the vaccinated animals and fewer metastases were observed than in the non-vaccinated animals. However, complete tumour remission was not achieved.
With regard to the prophylactic vaccination, the effect was even more significant: Here, the animals were at first vaccinated twice with the virus (days 0 and 21), and the cancer cells were administered twelve days later. The animals thus vaccinated were protected entirely from tumour development – no metastases could be identified.
“The findings have shown for the first time that the HERV-K envelope protein could be a useful target for the development of a vaccine, and may be able to provide new options for the treatment of various tumours” explained Professor Schnierle.
The Paul-Ehrlich-Institut, Federal Institute for Vaccines and Biomedicines in Langen in the federal state of Hessen, is a senior federal authority reporting to the Federal Ministry of Health (Bundesministerium für Gesundheit, BMG). It is responsible for the research, assessment, and marketing authorisation of biomedicines for human use and veterinary vaccines. Its remit also includes the authorisation of clinical trials and pharmacovigilance, i.e. recording and evaluation of potential adverse effects. Other duties of the institute include official batch control, scientific advice and inspections. In-house experimental research in the field of biomedicines and life science form an indispensable basis for the varied and many tasks performed at the institute. The PEI, with its roughly 800 staff, also has advisory functions at a national level (federal government, federal states (Länder)), and at an international level (World Health Organisation, European Medicines Agency, European Commission, Council of Europe etc.).
Weitere Informationen:
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0072756 Publication (Fulltext)
http://www.pei.de/EN/information/journalists-press/press-releases/2013/04-prehistoric-relics-targets-vaccination-against-cancer.html
Press Release on the PEI-Website
Media Contact
More Information:
http://www.pei.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















