New NIST method evaluates response to oxidation in live cells
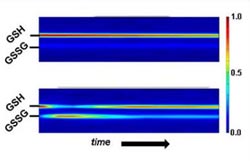
NMR data showing the levels of reduced glutathione (GSH) and oxidized glutathione (GSSG) over time in yeast cells at rest (top) and under oxidative stress (bottom). The cells at rest have almost no GSSG (dark blue shades) but it spikes briefly when they are stressed, while GSH declines. This type of analysis may one day help to measure cell health.<br><br>Credit: Reipa/NIST<br>
The NIST technique measures changes in a living cell's internal redox (reduction-oxidation) potential, a chemistry concept that expresses the favorability of reactions in which molecules or atoms either gain or lose electrons.
Redox reactions are important to cell chemistry because they regulate many genes and the proteins they produce. An accurate measure of redox potential can provide insight into how well these genes are working, and in turn, whether or not the activities they control—such as differentiation and growth—are functioning normally.
To assess this, scientists customarily measure the levels of both the reduced (electrons added) and oxidized (electrons lost) forms of glutathione, a peptide the cell uses as an antioxidant. Glutathione in cells is found predominately in the reduced state, known as GSH, but some gets converted to the oxidized form, known as GSSG. A high amount of GSSG indicates a cell has suffered oxidative stress, a process believed to contribute to cell aging, breakdown, malfunction (such as cancer) and eventual death.
Unfortunately, traditional methods of obtaining this data are akin to an autopsy. The only way to measure the relative amounts of GSH and GSSG within a cell has been to rupture its membrane—killing it—and then examine the released contents.
The NIST team developed a way to measure GSH and GSSG levels in living cells in real time using nuclear magnetic resonance (NMR)spectroscopy, a technique that images individual molecules similar to how doctors use magnetic resonance imaging (MRI) to noninvasively view organs. “NMR has been shown in recent years to be a powerful tool for studying metabolites as they operate in living cells, so we felt it could work well as a noninvasive way to do the same for GSH and GSSG,” says NIST research chemist Vytas Reipa.
In their proof-of-concept experiment,* the NIST researchers grew a mutant strain of yeast cells that could not manufacture their own glutathione in a medium containing the peptide tagged with a nitrogen isotope. This ensured that the only glutathione available in the cells would be detectable using NMR during its conversion from GSH to GSSG.
GSH and GSSG levels were measured by NMR for both cells at rest and under oxidative stress, and then used to calculate the changing intracellular redox potentials over time. The results showed, for the first time ever, that redox potential can serve as an indicator of how cells perform in response to oxidation in real time.
“We know that when oxidation tips the balance toward too much GSSG, we get a redox potential shifted more to the positive than it should be,” Reipa explains. “A healthy cell compensates by reversing the process and when that happens, the redox potential shifts back to its original value. A sick cell, on the other hand, does not compensate and the value stays positive. Therefore, an accurate in-cell measurement of redox potential could one day help us determine how well cells can recover from oxidative stress and, as a result, give us a picture of the cell's overall health.”
Currently, the NIST researchers are exploring other NMR-detectable peptides involved in reduction and oxidation processes to conduct studies with mammalian cells.
The NMR spectroscopy in this experiment was conducted at NIST's Gaithersburg, Md., facility in collaboration with NIST scientists at the Hollings Marine Laboratory (HML) in Charleston, S.C., and the Institute for Bioscience and Biotechnology Research (IBBR) in Rockville, Md.
*S.Y. Rhieu, A.A. Urbas, D.W. Bearden, J.P. Marino, K.A. Lippa and V. Reipa. Probing the intracellular glutathione redox potential by in-cell NMR spectroscopy. Angewandte Chemie, 126: 457-460 (2014). DOI: http://dx.doi.org/10.1002/anie.201308004.
Media Contact
More Information:
http://www.nist.govAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















