New Work Shines Light on Hox Genes Responsible for Firefly Lantern Development
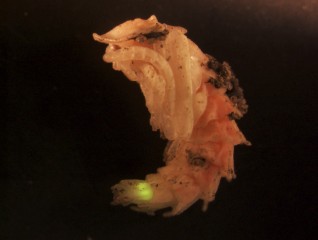
ARMIN MOCZEK AND MATTHEW STANSBURY A firefly's photic organ is functional throughout pupation and glows when the pupa is disturbed.
It’s difficult to identify a single evolutionary novelty in the animal kingdom that has fascinated and intrigued mankind more than the lantern of the firefly. Yet to this day, nothing has been known about the genetic foundation for the formation and evolution of this luminescent structure.
But now, new work from a former Indiana University Bloomington graduate student and his IU Ph.D. advisor offers for the first time a characterization of the developmental genetic basis of this spectacular morphological novelty — the firefly’s photic organ — and the means by which this beetle successfully uses ancient and highly conserved regulatory genes to form its lantern.
“Our study demonstrates unprecedented roles for two Hox genes,” said Armin Moczek, associate professor in the College of Arts and Sciences’ Department of Biology and co-author on the paper with his former Ph.D. student Matthew Stansbury, now a postdoc at the University of Arizona. “These genes have been highly conserved over very long evolutionary times to lay out the basic body forms of so many animals, from humans to flies. Our study now shows that on top of that, these Hox genes have recently acquired control over important aspects of lantern development.”
Hox genes are famous for specifying body regions and their boundaries, such as the head, thorax and abdomen of insects, or the cervical, thoracic and lumbar vertebrae of mammals. Hox genes are also famous for specifying which appendage will form on a given segment, such as a mouthpart in the head and a wing on the thorax, as well as specific modifications of these appendages: The beetle's forewing is a hardened shell rather than membranous due to the actions of a Hox gene, as is the reduction of the fly's hind wing to a tiny balancer organ.
What is nearly unprecedented, however, is that in the case of this beetle belonging to a family of about 2,000 species, the two Hox genes abdominal-A (abd-A) and Abdominal-B (Abd-B), appear to have acquired the ability to regulate an entirely novel, complex organ found nowhere else: the lantern.
“The notion that Hox genes can simply acquire new responsibilities quickly, especially without compromising old ones, is rather novel,” Moczek said. “This is one of relatively few examples that show that this indeed occurs in animal evolution.”
What Stansbury and Moczek found by down-regulating the function of each of these two genes was that both were required for proper adult photic organ formation.
“In particular, the Abd-B Hox gene was critical to all aspects of photic organ formation, suggesting it has evolved novel lantern-specific regulatory interactions,” Moczek said.
They also found that Abd-B repressed abdominal pigmentation in fireflies' photic organs, thereby creating a translucent cuticle that allows the light to escape the body interior. In addition, the sixth and seventh abdominal segments of most insects is ordinarily under the sole purview of abd-A rather than Abd-B.
However, it is precisely these segments that house the lantern in fireflies, suggesting the possibility of an anterior expansion of the domain of Abd-B function. That’s a big deal because Hox genes, given their hugely important functions in establishing the basic body plan, normally confine themselves to strict and highly conserved boundaries of expression.
“This is just the beginning,” Moczek said. “We want to know what are the target genes with which abd-A and Abd-B interact to form the organ; we want to get data on exactly when and where a given gene product is active; and we want to learn more about how the adult organ evolved from the larval organ, a much simpler structure located on yet another segment,” he added.
Moczek is no stranger to investigating novel traits, and the new work presents another example within his body of work that the possibility exists that Hox-gene mediated innovation — true, unique innovation — is more widespread than generally thought. Another example of this is beetle horns, one of Moczek’s primary areas of research: The horns of beetles are a novel trait that lack correspondence to other traits in beetles or insects. Work conducted by his former graduate student Bethany Wasik found that the positioning and growth of horns that come out of the thorax of beetles is regulated in part by the Hox gene Sex-combs-reduced.
And as novel traits are well-understood by Moczek, Hox genes are especially close to IU’s Department of Biology: The homeobox (Hox is the abbreviation) DNA sequence was discovered in 1983 by students in the laboratory of IU Distinguished Professor of Biology Thom Kaufman.
“This study is the first of its kind on these organisms and this structure, and we hope more than anything opens the door for future work on these charismatic insects,” Moczek said.
“The function of Hox and appendage-patterning genes in the development of an evolutionary novelty, the Photuris firefly lantern,” by Matthew Stansbury, University of Arizona, and Armin Moczek, Indiana University, was published today in the Proceedings of the Royal Society B.
Media Contact
More Information:
http://www.iu.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

How blue-green algae manipulate microorganisms
Research team at the University of Freiburg discovers previously unknown gene that indirectly promotes photosynthesis. Cyanobacteria – also called blue-green algae – are known as the “plants of the ocean”…

Combatting disruptive ‘noise’ in quantum communication
In a significant milestone for quantum communication technology, an experiment has demonstrated how networks can be leveraged to combat disruptive ‘noise’ in quantum communications. The international effort led by researchers…

Stretchable quantum dot display
Intrinsically stretchable quantum dot-based light-emitting diodes achieved record-breaking performance. A team of South Korean scientists led by Professor KIM Dae-Hyeong of the Center for Nanoparticle Research within the Institute for…





















