New technology for enzyme design
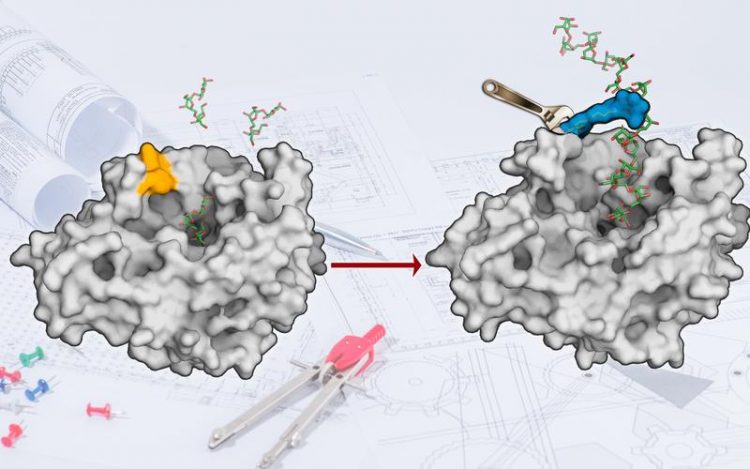
The surface of the enzyme levansucrase has been redesigned to produce sugar polymers. (Picture: AK Seibel)
Enzymes are tools of nature that accelerate almost all biochemical reactions in living cells as biological catalysts. For this reason, enzymes have been used in the chemical industry for some time now – in detergents and cleaners, toothpastes and shampoos, but also in foods. Enzymes help in the production of paper, textiles, leather, medicines, biofuels and other products.
Enzymes from the tailoring industry
Biochemically, enzymes are proteins that are composed of natural amino acids. They form a three-dimensional structure. Just like a key fits into a lock, each specific molecule fits into an enzyme and the enzyme converts it into a new product.
Technically, it is possible to exchange individual amino acids in an enzyme and thereby change its structure so that it can now process other molecules. In this way, British scientists have created just recently a mutant enzyme that breaks down plastic.
Surface of the levansucrase changed
Chemists from Julius-Maximilians-Universität Würzburg (JMU) in Bavaria, Germany, have now gone a step further in the tailoring of enzymes: “We thought what fascinating possibilities would arise if we could change the surface of enzymes chemically and use the chemical space of molecules,” says Jürgen Seibel , Professor of Organic Chemistry at the JMU. “We have developed a reaction that does not occur in nature in this way. It gives us a lot of freedom in reshaping enzyme surfaces. “
As the JMU scientists report in the journal “Chemical Science”, they first redesigned the surface of the enzyme levansucrase. Now, the enzyme can convert the table sugar (sucrose) directly into a polymer of fructose building blocks.
“So far, such a synthesis has been possible with levansucrase, but it works much more efficiently with the modified enzyme,” explains Seibel. The conversion of the enzyme per second is now significantly higher; moreover, it mainly produces the desired product and no accidental by-products.
Interesting for medicine and food industry
The fructose polymer could be used as a bio-gel for tissue transplantation in medicine or in the food industry – for example as a probiotic supplement in yogurts or baby food. Because like other functional sugars, the polymer could also serve certain intestinal bacteria as food and indirectly exert a health-promoting influence on the intestinal flora of humans.
“Product-Oriented Chemical Surface Modification of a Levansucrase (SacB) via an Ene-type Reaction”, Maria Elena Ortiz-Soto, Julia Ertl, Jürgen Mut, Juliane Adelmann, Thien Anh Le, Junwen Shan, Jörg Teßmar, Andreas Schlosser, Bernd Engels, Jürgen Seibel. Chemical Science 2018 Advance Article, DOI: 10.1039/C8SC01244J
Contact
Prof. Dr. Jürgen Seibel, Institute of Organic Chemistry, University of Würzburg, T +49 931 31-85326, seibel@chemie.uni-wuerzburg.de
Dr. Maria Ortiz-Soto, Institute of Organic Chemistry, University of Würzburg, T +49 931 3- 88733, ortiz.soto@uni-wuerzburg.de
Media Contact
More Information:
http://www.uni-wuerzburg.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















