MDC Researchers Gain New Insights into Epilepsy
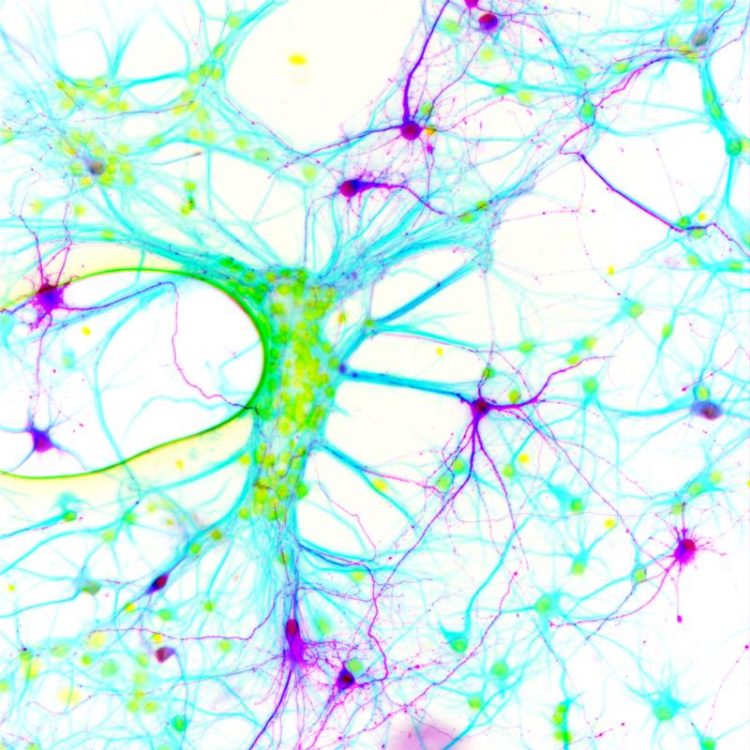
In order to identify the nerve cells in the neuronal network which express the altered glycine receptor researchers inserted an extra gene segment that illuminates the nerve cells (purple). (Photo: Jochen Meier/Copyright: MDC)
They regulate and coordinate the activity of groups of neurons that represent parts of information, which they then may transmit to other brain regions. If this precise system is thrown off balance, diseases can develop.
Professor Jochen Meier of the Max Delbrück Center (MDC) investigates such processes. Together with his colleagues, he has gained new insights into epilepsy, showing why the disease may present different symptoms (Journal of Clinical Investigation, doi:10.1172/JC171472)*.
The research focus is on the glycine receptor, known from previous studies in the field of spinal cord research as an inhibitory neurotransmitter receptor. Several years ago the research group of neurobiologists showed that this receptor is molecularly altered in patients with intractable temporal lobe epilepsy, one of the most common forms of epilepsy.
Expression of the altered receptor is increased in the hippocampus, a region of the brain that triggers seizures in most patients at later stages of the disease.
Professor Meier, who leads a Helmholtz junior research group at the explained that epilepsy is not exclusively inherited, that is, its cause need not be genetic. In fact, recent research data indicate that various environmental factors, including chronic stress, can cause epileptic seizures.
Resulting molecular and cellular mechanisms may change neuroplasticity to the extent that the affected tissue is no longer able to restore the normal state, which can then lead to chronic epilepsy. This may be one reason why the disease course differs from patient to patient.
The molecular alteration of the glycine receptor in the tissue of epilepsy patients is caused by a process known in research as “RNA editing”. Thereby, in the process of transcription of genetic information from DNA to RNA, individual letters are replaced with others. Enzymes are responsible for the editing of the text.
As a result, the original genetic text encoded in the DNA language no longer corresponds exactly to the RNA, which contains the code for the text building blocks of the proteins. The protein, in this case the glycine receptor, is altered so that it expresses a gain-of-function, and thus functions far more effectively than its non-edited counterpart.
Together with colleagues from Israel and different German universities, Dr. Aline Winkelmann and Professor Meier developed a new animal model of epilepsy to determine the role of this particular glycine receptor variant. This allowed them to express this receptor specifically in selected nerve cell types of the hippocampus and to investigate how it affects cognitive function and mood-related behavior.
They found that the RNA-edited gain-of-function receptor targets the presynaptic terminals, which transfer electrical impulses by releasing a neurotransmitter to other nerve cells. Thus, the function of selected types of nerve cells is strengthened, whereby the whole system of neuronal communication is thrown off balance.
Depending on whether excitatory or inhibitory nerve cells expressed the receptor, the mice were impaired in their cognitive abilities including memory deficits, or they showed increased anxiety.
“Our new animal model suggests that the same molecule can contribute to a wide range of symptoms in epilepsy patients – for example, cognitive dysfunction or anxiety – depending on which type of nerve cell expresses it,” Professor Meier said.
He and his colleagues have thus discovered a disease mechanism which they hope will open up new approaches to the development of targeted treatments for epilepsy patients. He stressed, however, “We must also identify the conductor of this cellular orchestra of dissonant molecular components that is responsible for receptor expression in varied nerve cells in the hippocampus of epilepsy patients.”
*Changes in neural network homeostasis trigger neuropsychiatric symptoms
Aline Winkelmann,1,2 Nicola Maggio,3 Joanna Eller,4 Gürsel Caliskan,5 Marcus Semtner,2 Ute Häussler,6 René Jüttner,7 Tamar Dugladze,4 Birthe Smolinsky,8 Sarah Kowalczyk,8 Ewa Chronowska,9 Günter Schwarz,8 Fritz G. Rathjen,7 Gideon Rechavi,10 Carola A. Haas,6,11 Akos Kulik,9,12 Tengis Gloveli,4,13 Uwe Heinemann,5 and Jochen C. Meier2
1FU-Berlin, Fachbereich Biologie, Chemie, Pharmazie, Berlin, Germany. 2RNA editing and Hyperexcitability Disorders Helmholtz Group, Max Delbrück Center for Molecular Medicine, Berlin, Germany. 3Talpiot Medical Leadership Program, Department of Neurology and the J. Sagol Neuroscience Center, The Chaim Sheba Medical Center, Tel HaShomer, Israel. 4Cellular and Network Physiology Group, Institute of Neurophysiology, Charité Universitätsmedizin Berlin, Berlin, Germany. 5CC2 Zentrum für Physiologie, Freie Universität Berlin, Berlin, Germany. 6Experimental Epilepsy Research, Department of Neurosurgery, Neurocenter, University of Freiburg, Freiburg, Germany. 7Developmental Neurobiology, Max Delbrück Center for Molecular Medicine, Berlin, Germany. 8Institute of Biochemistry, University of Cologne and Center for Molecular Medicine, Cologne, Germany. 9Department of Physiology II, University of Freiburg, Freiburg, Germany. 10Sheba Cancer Research Center, The Chaim Sheba Medical Center and Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel. 11BrainLinks-BrainTools, Cluster of Excellence and 12BIOSS Centre for Biological Signalling Studies, University of Freiburg, Freiburg, Germany. 13Bernstein Center for Computational Neuroscience Berlin, Berlin, Germany.
Contact:
Barbara Bachtler
Press Department
Max Delbrück Center for Molecular Medicine (MDC) Berlin-Buch
in the Helmholtz Association
Robert-Rössle-Straße 10
13125 Berlin
Germany
Phone: +49 (0) 30 94 06 – 38 96
Fax: +49 (0) 30 94 06 – 38 33
e-mail: presse@mdc-berlin.de
http://www.mdc-berlin.de/
http://www.mdc-berlin.de/1158081/en/research/research_teams/rna_editing_and_hype…
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Silicon Carbide Innovation Alliance to drive industrial-scale semiconductor work
Known for its ability to withstand extreme environments and high voltages, silicon carbide (SiC) is a semiconducting material made up of silicon and carbon atoms arranged into crystals that is…

New SPECT/CT technique shows impressive biomarker identification
…offers increased access for prostate cancer patients. A novel SPECT/CT acquisition method can accurately detect radiopharmaceutical biodistribution in a convenient manner for prostate cancer patients, opening the door for more…

How 3D printers can give robots a soft touch
Soft skin coverings and touch sensors have emerged as a promising feature for robots that are both safer and more intuitive for human interaction, but they are expensive and difficult…





















