Keeping traffic moving
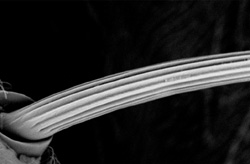
Figure 1: An electron microscope image of a sensory bristle from the body of the fruit fly Drosophila melanogaster. Copyright : 2011 Tetsuhisa Otani <br>
The body of the adult fruit fly is covered with hair-like bristles (Fig. 1) that act as sensory organs for detecting tactile stimuli. Each one consists of a single cell that has gradually elongated over the course of pupal development, reinforced by bundles of actin protein filaments.
The signaling protein IKKå helps to regulate this process by controlling the organization of these actin bundles, but a recent study from Shigeo Hayashi and colleagues at the RIKEN Center for Development Biology in Kobe has revealed that IKKå also promotes bristle growth by managing the trafficking of cellular cargoes (1).
Initial experiments by Hayashi and team showed that activated IKKå is primarily found at the tips of developing bristles, where growth-associated cargoes are most likely to be unloaded. “Membranes and associated proteins are water-insoluble and thus do not easily diffuse to distant sites, and one model is that distal trafficking actively delivers such insoluble materials as packages,” explains Hayashi.
Membrane-enclosed bubbles known as endosomes are a core component in this process, using so-called motor proteins to travel along routes defined by a microscopic ‘railway’ of fibers known as microtubules. The researchers found that this trafficking is severely disrupted in the absence of IKKå, with endosomes remaining trapped at the ends of the bristle rather than being distributed throughout the cell.
Hayashi and colleagues determined that IKKå interacts with a protein called Nuf, which links the motor protein Dynein with a key endosome-associated protein and thus contributes to directional transport of cargoes toward the tip of the growing bristle. Upon arrival at the tip, IKKå-mediated inactivation of Nuf sends the newly emptied endosomes on a return trip, thereby completing a ‘recycling’ process. “Such endosomal movement occurs in other cell types, but the shape of bristles makes this shuttling very prominent,” says Hayashi. “I think this is a very good example of how a highly specialized cell and its shape can reveal a mechanism of general significance.”
Many other cells grow in a similar fashion, ranging from the tiny branches that help connect neurons to the hairs on plant roots that assist in water absorption, and Hayashi speculates that similar regulatory mechanisms may also operate in these contexts. Moving forward, he and his colleagues will further explore the apparently central coordinating role of IKKå. “We are currently studying actin as a target,” says Hayashi, “and we are also studying upstream regulators of IKKå, hoping to uncover a comprehensive view of this signaling pathway.”
The corresponding author for this highlight is based at the Laboratory for Morphogenetic Signaling, RIKEN Center for Developmental Biology
Journal information
(1) Otani, T., Oshima, K., Onishi, S., Takeda, M., Shinmyozu, K., Yonemura, S. & Hayashi, S. IKKå regulates cell elongation through recycling endosome shuttling. Developmental Cell 20, 219–232 (2011).
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Silicon Carbide Innovation Alliance to drive industrial-scale semiconductor work
Known for its ability to withstand extreme environments and high voltages, silicon carbide (SiC) is a semiconducting material made up of silicon and carbon atoms arranged into crystals that is…

New SPECT/CT technique shows impressive biomarker identification
…offers increased access for prostate cancer patients. A novel SPECT/CT acquisition method can accurately detect radiopharmaceutical biodistribution in a convenient manner for prostate cancer patients, opening the door for more…

How 3D printers can give robots a soft touch
Soft skin coverings and touch sensors have emerged as a promising feature for robots that are both safer and more intuitive for human interaction, but they are expensive and difficult…





















