Jacobs researchers develop new carbene complexes for versatile industrial application
New compounds with liquid-crystalline characteristics<br>Photo: Jacobs University<br>
N-Heterocyclic carbenes (NHC) in particular were soon used in pharmaceutical and technical processes. Scientists at Jacobs University have now developed new ways of synthesizing stable NHC-complexes of main-group elements, which are user-friendly. They can be used as chemical overcharge protection in lithium ion batteries or possibly as liquid crystals in LC-displays.
The German Research Foundation, the Federal Ministry of Economics and Technology and the Merck KGaA support the research with a total of €1.2million.
Starting from the precursor DFI (1,3-dimethyl-2,2-difluoroimidazolidine) Gerd-Volker Röschenthaler, Professor of Chemistry at Jacobs University and Dr. Tobias Böttcher for the first time produced a carbene complex of phosphor(V) through oxidative addition to PF3 and later from easier-to handle PCl3.
“Our aim was to find synthetic possibilities, which are easy to apply and which could simplify processes in research and development,” says Prof. Röschenthaler.
The new carbene complex NHC-PF5 allows for a variety of practical applications. In cooperation with Prof. Martin Winter from the MEET Institute at the University of Münster the Jacobs researchers learned that NHC-PF5 could be effective as chemical overcharge protection in lithium ion batteries: By adding the complex to its electrolyte components, the battery deactivated at 4.6 volt. An overcharge causing the battery to burst, ignite or explode can be prevented. The scientists are now looking into the reasons why NHC-PF5 serves well as an overcharge protection. Their collaborative research is supported by the Federal Ministry of Economics and Technology as well as industrial partners – among them BASF, Volkswagen AG, SGL Carbon, Toda Europe and Merck. Together with the University of Münster a patent has been filed for “Electrolyte additive for lithium-based energy sources” (University of Münster, Jacobs University Bremen), DE 10 2011 055 028.3, PCT/EP2012/071544).
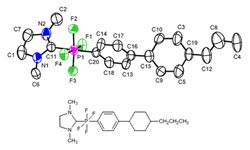
In cooperation with Dr. Matthias Bremer from Merck, Prof. Röschenthaler, Dr. Böttcher and their team (Dr. Romana Pajkert and Dr. Maksym Ponomarenko) were also able to develop new compounds with liquid-crystalline characteristics. The compounds (see image of molecular structure) contain exceptionally large dipole moments, a prerequisite for application as liquid crystals. This particular class of compounds could constitute a new form of liquid crystals. The dipole moment serves to improve their properties, which in turn could lead to increased brilliance in smartphone displays or faster circuit times in monitors and flat-screens.
Together with Prof. Berthold Hoge from University of Bielefeld the Jacobs researchers could show that phosphor(V) carbene complexes could be split neatly by hydrogen fluoride to chemical salts with a very low melting point (they are liquid at room temperature). They can be used widely spread as so-called ionic liquids. Being easily produced in just one step the new synthetic route is of great interest to chemical industry as reaction medium in organic syntheses.
Another way to synthesize carbene complexes Röschenthaler and Böttcher found in carbene transfer: an inexpensive, simple and highly versatile method. First a carbene complex is synthesized that is both stable and at the same time labile enough to transfer the carbene to the favored element. The scientists used NHC-SiCl4 as transfer reagent and transferred the carbene to PCl3 (Chemical Science, 2013, 4, 77-83, DOI: 10.1039/C2SC21214E). This is the first time a carbene has been transferred from one main group element to another. Furthermore, it is also possible to transfer carbenes to transition metals, which the scientists could show with nickel(II) and palladium(II)chloride. Carbene complexes of these metals often show good catalytic properties and thus are relevant for industry. A patent has been filed for this new manufacturing method as well: „Silicon-based Carbene Transfer Reagent“ (Jacobs University Bremen, DE 10 2012 102 535.5).
Contact:
Gerd-Volker Röschenthaler | Professor of Chemistry
E-mail: g.roeschenthaler@jacobs-university.de | Phone: +49 421 200-3138
Media Contact
More Information:
http://www.jacobs-university.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















