Hide and Seek: Revealing Camouflaged Bacteria
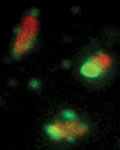
GTPases (green) attack Salmonella typhimurium (red). (Figure: University of Basel, Biozentrum)
Bacteria have developed countless strategies to hide themselves in order to evade attack by the immune system. In the body, Salmonella bacteria use macrophages as host cells to ensure their survival and to be able to spread within the body.
Their survival strategy is to nestle into a vacuole within the cytoplasm of a macrophage, hiding there and multiplying. While they are hidden there, the immune cells cannot detect the bacteria and fight them.
The macrophages, in which the Salmonella hide, however, have also developed a strategy to unmask the disguise of the bacterium and uncover its hiding place. Prof. Petr Broz’s research group at the Biozentrum of the University of Basel has discovered a protein family called interferon-induced GTPases in host cells invaded by Salmonella.
“They are responsible for destroying the hiding place of the pathogen and to initiate the immune response of the cell,” explains Etienne Meunier, first author of the publication.
Once the hiding place has been discovered, GTPases are transported to the vacuole and destabilize its membrane. The bacteria are left behind unprotected in the cytoplasm where their surface molecules are easily recognized by the intracellular defense.
“The GTPases are the key to the hiding place of the bacteria. Once the door has been opened and the protective vacuole destroyed, there is no escape. The bacteria are immediately exposed to the defense machinery of the cell”, says Meunier. Receptors in the cell identify the pathogen, which then activate special cellular enzymes to destroy the bacteria. In addition, the cells own proteases, so-called caspases, are activated and trigger cell death of the infected host cell.
Salmonella still remain a feared pathogenic agent, as they can cause life threatening diarrheal disease. The findings of Broz and his team enable the better understanding of the strategies of the immune cells and to perhaps model this in the future.
The deeper understanding of the immune response of our cells also paves the way for new approaches in using drugs to support the body’s fight against pathogens. In order to further elucidate the mechanisms of the immune response to Salmonella infections, the research team plans to investigate how cells detect the hiding place of the bacteria, the vacuole in the cytoplasm of the macrophages, and what initiates the recruitment of GTPases to the vacuole.
Original Citation
Etienne Meunier, Mathias S. Dick, Roland F. Dreier, Nura Schürmann, Daniela Kenzelmann Broz, Søren Warming, Merone Roose-Girma, Dirk Bumann, Nobuhiko Kayagaki, Kiyoshi Takeda, Masahiro Yamamoto and Petr Broz
Caspase-11 activation requires lysis of pathogen-containing vacuoles by IFN-induced GTPases
Nature (2014); Advance Online Publication | doi: 10.1038/nature13157
Further Information
Prof. Petr Broz, University of Basel, Biozentrum, phone: +41 61 267 23 42, email: petr.broz@unibas.ch
Media Contact
More Information:
http://www.unibas.chAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…
Innovative microscopy demystifies metabolism of Alzheimer’s
Researchers at UC San Diego have deployed state-of-the art imaging techniques to discover the metabolism driving Alzheimer’s disease; results suggest new treatment strategies. Alzheimer’s disease causes significant problems with memory,…
A cause of immunodeficiency identified
After stroke and heart attack: Every year, between 250,000 and 300,000 people in Germany suffer from a stroke or heart attack. These patients suffer immune disturbances and are very frequently…





















