Head in a Cage
<br>
In the human body, lipids do not only serve as energy stores and structural elements, but they are also important signaling molecules. Disruptions of lipid signal transmission seem to be involved in diseases such as atherosclerosis and diabetes, as well as inflammation and pain.
In the journal Angewandte Chemie, researchers from Heidelberg have now reported on photoactivatable lipids that can be used to manipulate signaling processes in cells with both spatial and time resolution.
To communicate with each other and react to external stimuli, cells need signal-transmission mechanisms. The signal cascades involved are very complex and vary greatly from one cell type to the next. For example, one type of cascade involves the activation of phospholipase C, which then splits a membrane building block into inositol trisphosphate and the lipid diacylglycerol (DAG). These in turn serve as secondary messengers within the cell.
DAG anchors the enzyme protein kinase C (PKC) to the cell membrane and activates it. In addition, DAG can open certain calcium channels in the cell membrane, allowing calcium ions to flow into the cell. This stimulates further steps of the network and hence eventually trigger physiological responses, such as changes in gene expression.
Lipids as secondary messengers have received relatively little attention from researchers. Lipids consist of a head group and a “tail” made of a hydrocarbon chain that can vary greatly with regard to its length and the number, distribution, and arrangement of its double bonds. Previous investigations did not differentiate the effects of the different tails, only those of the heads.
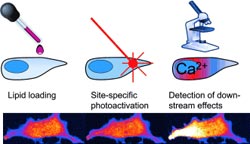
Carsten Schultz and a team from the European Molecular Biology Laboratory (EMBL) in Heidelberg have now taken on this challenge. They synthesized DAG lipids with a variety of tails and locked their glycerol heads into “cages” – molecules attached so as to block and deactivate the head group.
The cages are designed with a built-in “break-away” point that breaks open upon irradiation with light to release the DAG. These types of photoactivatable molecules make it possible to deliver biologically active signal molecules at a specific time and place with subcellular resolution.
Through these experiments, the researchers were able to demonstrate that the activation of PKC is locally limited by DAG, whereas the elevation of internal calcium ion concentration through activation of calcium channels affects the entire cell. Surprisingly, these effects seem to be dependent on the fatty acid composition of the lipid. One of the DAG variants thus induced fewer, shorter, and weaker elevations of the calcium level, while another caused stronger, long-lasting calcium signals. A third had no significant influence on the intracellular calcium concentration.
“If this variability concerning the fatty acid composition should influence the control of cellular processes in most lipids, a completely new level of complexity has to be considered in cell biology”, says Schultz. “Furthermore, our results demonstrate that cells can respond to a given spatially confined signal both with a local and a global response pattern. Local signaling is particularly important in polarized and migrating cells, where different signals are needed at opposite ends of the cell.”
About the Author
Dr Carsten Schultz is an interdisciplinary team leader and Senior Scientist at the European Molecular Biology Laboratory (EMBL) in Heidelberg. His main research fields include the manipulation and visualization of intracellular signal transduction networks by using chemical biology, as well as the development of fluorescent detection systems, for example in applications concerning chronic lung disorders within the framework of the German Center for Lung Research (DZL) in Heidelberg.
Author: Carsten Schultz, European Molecular Biology Laboratory, Heidelberg (Germany), http://www.embl.de/research/units/cbb/schultz/members/index.php?s_personId=CP-60002438
Title: The Fatty Acid Composition of Diacylglycerols Determines Local Signaling Patterns
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201301716
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















