Gene therapy for hearing loss: potential and limitations
<br>
Regenerating sensory hair cells, which produce electrical signals in response to vibrations within the inner ear, could form the basis for treating age- or trauma-related hearing loss. One way to do this could be with gene therapy that drives new sensory hair cells to grow.
Researchers at Emory University School of Medicine have shown that introducing a gene called Atoh1 into the cochleae of young mice can induce the formation of extra sensory hair cells.
Their results show the potential of a gene therapy approach, but also demonstrate its current limitations. The extra hair cells produce electrical signals like normal hair cells and connect with neurons. However, after the mice are two weeks old, which is before puberty, inducing Atoh1 has little effect. This suggests that an analogous treatment in adult humans would also not be effective by itself.
The findings were published May 9 in the Journal of Neuroscience.
“We’ve shown that hair cell regeneration is possible in principle,” says Ping Chen, PhD, associate professor of cell biology at Emory University School of Medicine. “In this paper, we have identified which cells are capable of becoming hair cells under the influence of Atoh1, and we show that there are strong age-dependent limitations on the effects of Atoh1 by itself.”
The first author of the paper, Michael Kelly, now a postdoctoral fellow at the National Institute on Deafness and Other Communication Disorders, was a graduate student in Emory’s Neuroscience program.
Kelly and his coworkers engineered mice to turn on the Atoh1 gene in the inner ear in response to the antibiotic doxycycline. Previous experimenters had used a virus to introduce Atoh1 into the cochleae of animals. This approach resembles gene therapy, but has the disadvantage of being slightly different each time, Chen says. In contrast, the mice have the Atoh1 gene turned on in specific cells along the lining of the inner ear, called the cochlear epithelium, but only when fed doxycycline.
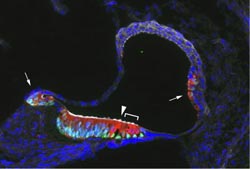
Young mice given doxycycline for two days had extra sensory hair cells, in parts of the cochlea where developing hair cells usually appear, and also additional locations (see accompanying image).
The extra hair cells could generate electrical signals, although those signals weren’t as strong as mature hair cells. Also, the extra hair cells appeared to attract neuronal fibers, which suggests that those signals could connect to the rest of the nervous system.
“They can generate electrical signals, but we don’t know if they can really function in the context of hearing.” Chen says. “For that to happen, the hair cells’ signals need to be coordinated and integrated.”
Although doxycycline could turn on Atoh1 all over the surface of the cochlea, extra sensory hair cells did not appear everywhere. When they removed cochleae from the mice and grew them in culture dishes, her team was able to provoke even more hair cells to grow when they added a drug that inhibits the Notch pathway.
Manipulating the Notch pathway affects several aspects of embryonic development and in some contexts appears to cause cancer, so the approach needs to be refined further. Chen says that it may be possible to unlock the age-related limits on hair cell regeneration by supplying additional genes or drugs in combination with Atoh1, and the results with the Notch drug provide an example.
“Our future goals are to develop approaches to stimulate hair cell formation in older animals, and to examine functional recovery after Atoh1 induction,” she says.
The research was supported by the National Institute on Deafness and Other Communications Disorders, the National Basic Research Program of China and the Natural Science Foundation of China.
Reference: M.C. Kelly, Q. Chang, A. Pan, X. Lin and P. Chen. Atoh1 directs the formation of sensory mosaics and induces cell proliferation in the postnatal mammalian cochlea in vivo. J. Neurosci. 32: 699-6710 (2012).
Media Contact
More Information:
http://www.emory.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















