Fly Cells Flock Together, Follow the Light

The study of this unexpected cell movement, reported May 16 in Nature Cell Biology, holds potential importance for understanding embryonic development, wound healing and tumor metastasis — the process by which tumor cells acquire the ability to invade surrounding tissues and migrate long distances to colonize lymph nodes, bones and other distant organs.
The research dramatically demonstrates, the researchers say, the collective direction-sensing behavior of live cells in intact tissue, and a means of controlling protein behavior in a living organism by shining a focused beam of light precisely on the parts of cells where they want the protein to be active.
“Our little system in the fruit fly is an elegant example of cells behaving socially in their natural environment — surrounded by other cells,” says Denise Montell, Ph.D., a professor of biological chemistry and director of the Center for Cell Dynamics at the Johns Hopkins University School of Medicine. “You can’t capture this behavior if you look at individual cells in a culture dish.”
The “social” migrating behavior among a cluster of cells in the fly ovary surprised the research team, which was using a new laser light tool to manipulate protein activity.
“People tend to think of cancer as single cells breaking off from the tumor and migrating away,” Montell says, but it’s likely that this collective form of movement is important, at one phase or another, in the spread of tumors.”
A better understanding of how and why cells move can facilitate the development of new treatments not only for cancer but other disorders characterized by aberrant cell behavior.
Developed in the laboratory of Klaus Hahn, Ph.D., Thurman Professor of Pharmacology at the University of North Carolina at Chapel Hill, the light-activation technique previously had been shown to control cell movement in cultured mammalian cells. The Hopkins-led study provides proof of principle that a non-toxic light alone can activate a protein in live organisms, allowing researchers to safely control when and where cells move.
The Hopkins team conducted their study on a cluster of six so-called border cells in the fly ovary, cells the team has long studied and which are important to the fly because if they don’t migrate, females are sterile. In addition they serve as a model for understanding the mechanisms that control collective cell movements in general, which occur during normal embryonic development, wound healing and in tumor metastasis.
First, they genetically altered the border cells so that they were lacking the ability to respond to naturally occurring chemical attractants that normally control their movement. Then they used a fly protein known as Rac, which was fused to a photoactivatable (PA) plant protein, a creation engineered by Hahn’s lab. The PA-Rac, which remains inert in the dark, reacts to light because the plant protein changes shape and allows Rac to become active, causing the cells to move.
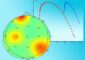
Because a beam of laser light can be much smaller than a cell, the team was able to activate Rac not only in one single cell, but also in one part of one cell, Montell says: “The other cool thing is this is reversible, so as soon as you take the light away, the PA-Rac wraps back up and turns itself off.”
Following up on previous research, the team wanted to find out if Rac would be sufficient to set the direction of movement of cells within live tissue.
When they shined a laser beam on various individual cells, the entire cluster responded by moving in directions that it wouldn’t under normal conditions: sideways, for instance, and even in reverse. In short, they followed the light.
“When we activated Rac in even one part of one of these cells — and not in the cell that would be the leader if all was normal — it was as if all the other cells said, Aha! You’ve got more Rac activity so we’re heading your way,” Montell says. “It’s amazing to me that somehow the cells sense each others’ levels of Rac activity and collectively decide which way to go.
Authors on the paper, in addition to Montell and Hahn, are Xiaobo Wang from Johns Hopkins and Yi Wu from UNC.
Funding was provided by the National Institutes of Health and the Cell Migration Consortium.
On the Web:
A video clip of a photo-activatable form of Rac is available at: http://www.icm.com/montell/MovieS5%28RacQ61Lforwardandrev%29.mov
Denise Montell Lab: http://www.hopkinsmedicine.org/dmontell/
Nature Cell Biology: http://www.nature.com/ncb/index.html
Cell Migration Consortium: http://www.cellmigration.org/index.shtml
Media Contact
More Information:
http://www.jhmi.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Security vulnerability in browser interface
… allows computer access via graphics card. Researchers at Graz University of Technology were successful with three different side-channel attacks on graphics cards via the WebGPU browser interface. The attacks…

A closer look at mechanochemistry
Ferdi Schüth and his team at the Max Planck Institut für Kohlenforschung in Mülheim/Germany have been studying the phenomena of mechanochemistry for several years. But what actually happens at the…

Severe Vulnerabilities Discovered in Software to Protect Internet Routing
A research team from the National Research Center for Applied Cybersecurity ATHENE led by Prof. Dr. Haya Schulmann has uncovered 18 vulnerabilities in crucial software components of Resource Public Key…