Crystal structure of archael chromatin clarified in new study
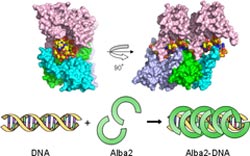
Polimerization of Alba2-DNA complex structure<br>Copyright : RIKEN<br>
Three distinct evolutionary branches of organisms make up all natural forms of life on the planet: bacteria, archaea and eukaryotes. Among these three, the domain known as archaea includes a variety of organisms that live in harsh environments similar to those of an early Earth, thus offering arguably the greatest glimpse of what life may have looked like 4 billion years ago.
One area of great interest is the process by which DNA bind to proteins to compact and regulate the availability of genetic material, a process which is essential in all cellular organisms. In eukaryotes, proteins known as “histones” package and order DNA into a compact protein-DNA structure called chromatin. Archaea, in contrast, have no such universal chromatin proteins, instead using two or more DNA-binding proteins to package DNA. Alba is the most widespread and abundant such archaeal chromatin protein, present in the genome sequence of every archaeal species that lives in high-temperature environments (thermophilic or hyperthermophilic).
While researchers know about the existence of Alba in archaea, the question of how these proteins bind to and compact DNA has remained a mystery. To answer this question, the researchers analyzed the crystal structure of the Alba2-DNA complex from the archaea A. pernix K1 at atomic-level resolution using synchrotron radiation from the RIKEN SPring-8 facility in Harima, Japan. Their results indicate that unlike the chromatin structure of eukaryotes, Alba2 in archaea forms a hollow pipe with the duplex DNA running through it, with the hairpin structure of Alba2 stabilizing the pipe.
Published in the February 10th issue of the Journal of Biological Chemistry, this newly-discovered mechanism for compacting DNA marks a major step forward in our understanding of the evolution of chromatin structure. The results promise to clarify how abnormalities in chromatin structure can contribute to cancers and gene disorders, while also providing inspiration for the development of new types of biodevices.
Contact:
Thirumananseri Kumarevel
Biometal Science Laboratory
RIKEN SPring-8 Center
Tel: +81-(0)791-58-0802 / Fax: +81-(0)791-58-2917
Global Relations Office
RIKEN
Tel: +81-(0)48-462-1225 / Fax: +81-(0)48-463-3687
Mail: koho@riken.jp
Reference:
Tomoyuki Tanaka, Sivaraman Padavattan and Thirumananseri Kumarevel “Crystal structure of archael chromatin protein Alba2-dsDNA complex from Aeropyrum pernix K1.” Journal of Biological Chemistry, 2012, DOI: 10.1074/jbc.M112.343210
About RIKEN
RIKEN is Japan's flagship research institute devoted to basic and applied research. Over 2500 papers by RIKEN researchers are published every year in reputable scientific and technical journals, covering topics ranging across a broad spectrum of disciplines including physics, chemistry, biology, medical science and engineering. RIKEN's advanced research environment and strong emphasis on interdisciplinary collaboration has earned itself an unparalleled reputation for scientific excellence in Japan and around the world.
About the RIKEN SPring-8 Center
The RIKEN SPring-8 Center, located in Harima, Japan, is home to SPring-8 (the Super Photon ring-8 GeV), the most powerful synchrotron radiation facility in the world. The RIKEN SPring-8 Center was established to support frontier research initiatives applying SPring-8's unique radiation to a wide variety of fields, notably structural biology and materials science. The center also focuses on the development of technology for producing high-quality synchrotron radiation sources and on development of the new SACLA X-ray Free Electron Laser project.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















