Complex neural circuitry keeps you from biting your tongue
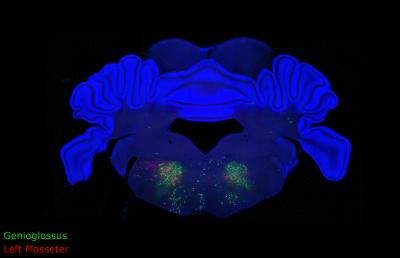
In this blue cross-section of a mouse brain, two colors of fluorescent dye trace the premotor neurons that close the jaw and stick out the tongue, revealing how the brain is wired to coordinate these muscles during chewing, drinking, and vocalizing. Credit: Fan Wang Lab, Duke University
Eating, like breathing and sleeping, seems to be a rather basic biological task. Yet chewing requires a complex interplay between the tongue and jaw, with the tongue positioning food between the teeth and then moving out of the way every time the jaw clamps down to grind it up. If the act weren't coordinated precisely, the unlucky chewer would end up biting more tongue than burrito.
Duke University researchers have used a sophisticated tracing technique in mice to map the underlying brain circuitry that keeps mealtime relatively painless. The study, which appears June 3 in eLife, could lend insight into a variety of human behaviors, from nighttime teeth grinding to smiling or complex vocalizations.
“Chewing is an activity that you can consciously control, but if you stop paying attention these interconnected neurons in the brain actually do it all for you,” said Edward Stanek IV, lead study author and graduate student at Duke University School of Medicine. “We were interested in understanding how this all works, and the first step was figuring out where these neurons reside.”
Previous mapping attempts have produced a relatively blurry picture of this chewing control center. Researchers know that the movement of the muscles in the jaw and tongue are governed by special neurons called motoneurons and that these are in turn controlled by another set of neurons called premotor neurons. But the exact nature of these connections — which premotor neurons connect to which motoneurons — has not been defined.
Senior study author Fan Wang, Ph.D., associate professor of neurobiology and a member of the Duke Institute for Brain Sciences, has been mapping neural circuits in mice for many years. Under her guidance, Stanek used a special form of the rabies virus to trace the origins of chewing movements.
The rabies virus works naturally by jumping backwards across neurons until it has infected the entire brain of its victim. For this study, Stanek used a genetically disabled version of rabies that could only jump from the muscles to the motoneurons, and then back to the premotor neurons. The virus also contained a green or red fluorescent tag, which enabled the researchers to see where it landed after it was done jumping.
Stanek injected these fluorescently labeled viruses into two muscles, the tongue-protruding genioglossus muscle and the jaw-closing masseter muscle. He found that a group of premotor neurons simultaneously connect to the motoneurons that regulate jaw opening and those that trigger tongue protrusion.
Similarly, he found another group that connects to both motoneurons that regulate jaw closing and those responsible for tongue retraction. The results suggest a simple method for coordinating the movement of the tongue and jaw that usually keeps the tongue safe from injury.
“Using shared premotor neurons to control multiple muscles may be a general feature of the motor system,” said Stanek. “For other studies on the rest of the brain, it is important to keep in mind that individual neurons can have effects in multiple downstream areas.”
The researchers are interested in using their technique to jump even further back in the mouse brain, eventually mapping the circuitry all the way up to the cortex. But first they plan to delve deeper into the connections between the premotor and motoneurons.
“This is just a small step in understanding the control of these orofacial movements,” Stanek said. “We only looked at two muscles and there are at least 10 other muscles active during chewing, drinking, and speech. There is still a lot of work to look at these other muscles, and only then can we get a complete picture of how these all work as a unit to coordinate this behavior,” said Stanek.
The research was supported by grants from the National Institutes of Health (NS077986 and DE019440).
CITATION: “Monosynaptic Premotor Circuit Tracing Reveals Neural Substrates for Oro-motor Coordination,” Edward Stanek IV, Steven Chang, Jun Takatoh, Bao-Xia Han, and Fan Wang. eLife, June 3, 2014. DOI: 10.7554/eLife.02511 http://elifesciences.org/content/early/2014/04/30/eLife.02511
Media Contact
More Information:
http://duke.edu/All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…





















