Color without Dyes: New non-iridescent structural colors cover the whole spectrum
<br>
The researchers reveal the secret of their success in the journal Angewandte Chemie: their “photonic pigments” are microcapsules filled with densely packed core–shell colloidal particles.
Conventional coloring agents have a variety of disadvantages: organic dyes tend to fade; inorganic pigments are often based on toxic heavy metals such as chromium. The color we see results from the absorption of a portion of the visible light spectrum. The reflected portions add to the color observed.
Another way to produce color that works without absorption is widely found in nature – in butterflies, for example. Arrays of nanoscopic particles can appear to be colored as a result of wavelength-dependent optical interference, refraction, and light scattering.
The color depends on the size of the particles. However, such structural pigments iridesce, meaning that the observed color varies in accordance with the angle of illumination or the viewing angle. In displays and many other applications, this would naturally be very annoying. The high degree of order in the particles of the crystal lattice contributes to this problem.
It is thus desirable to have the particles in a noncrystalline, amorphous arrangement, which is very difficult to achieve. In addition, amorphous structural pigments have thus far had very unsatisfactory color saturation caused by so called multiple scattering. A second type of undesired scattering, so-called incoherent scattering, contributes to a blueish background color that makes it difficult to produce a full spectrum of colors, particularly red.
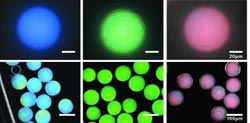
A team from Harvard University (USA), the Korea Advanced Institute of Science and Technology, and the Korea Electronics Technology Institute has now solved these problems. Their success is due to microcapsules packed with nanoscopic polymer spheres whose core and shell are made of two different polymers. Led by Vinothan N. Manoharan, the scientists designed the shells to have the same refractive index as the surrounding aqueous medium.
The light is thus only scattered by the cores, whose size and distance from each other determine the scattering properties. In a dense packing arrangement, the distance between cores can be determined by the thickness of the shells. If the cores are very small and the shells relatively thick, the undesired types of scattering can be minimized while the desired coherent scattering that is responsible for the structural color dominates.
By using a microfluidic technique, tiny droplets of an aqueous suspension of the core–shell particles are coated with a thin film of oil. They are then shrunken through osmosis until the particles adopt a densely packed arrangement. The soft polymer shells of the particles prevent crystallization. In the last step, the oil film is cured with UV light to make delicate, transparent, capsules.
The color of the novel structural pigments can be varied over the entire spectrum by changing the distances of the particle cores from each other by means of the thickness of the shells. The goal is to use these new nanoparticles in reflective displays.
About the Author
Vinothan N. Manoharan is the Gordon McKay Professor of Chemical Engineering and Professor of Physics at Harvard University. His research focuses on understanding the physics of self-assembly and controlling self-assembly processes to make interesting materials.
Author: Vinothan N. Manoharan, Harvard University, Cambridge (USA), http://manoharan.seas.harvard.edu/people.html
Title: Full-Spectrum Photonic Pigments with Non-iridescent Structural Colors through Colloidal Assembly
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201309306
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A new look at the consequences of light pollution
GAME 2024 begins its experiments in eight countries. Can artificial light at night harm marine algae and impair their important functions for coastal ecosystems? This year’s project of the training…

Silicon Carbide Innovation Alliance to drive industrial-scale semiconductor work
Known for its ability to withstand extreme environments and high voltages, silicon carbide (SiC) is a semiconducting material made up of silicon and carbon atoms arranged into crystals that is…

New SPECT/CT technique shows impressive biomarker identification
…offers increased access for prostate cancer patients. A novel SPECT/CT acquisition method can accurately detect radiopharmaceutical biodistribution in a convenient manner for prostate cancer patients, opening the door for more…





















