Cellular traffic control system mapped for the first time
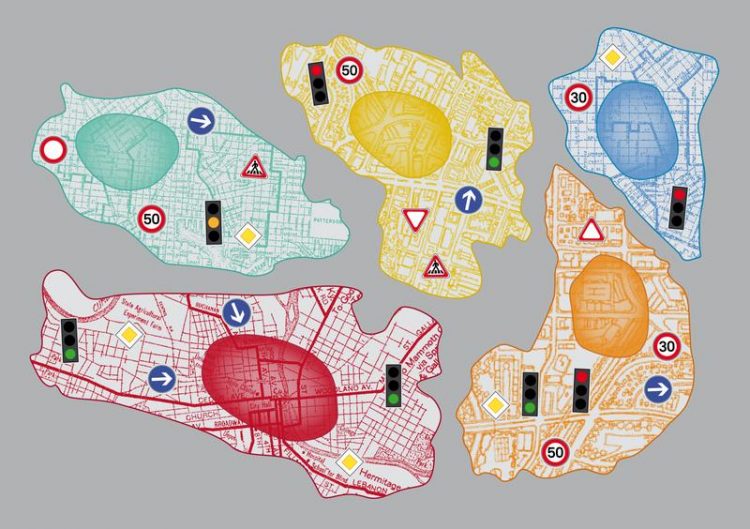
The figure illustrates singles cells as city with intracellular roads. Traffic lights and signs necessary to guide the traffic flow are highlighted. picture: Sarah Steinbacher/UZH
Cells must transport nutrients and messenger cargos through its membrane and transport them within the cell at the correct time and place. This procedure is complex and is regulated with the help of specific genes. If disturbances in the transport mechanism arise, severe diseases, such as diabetes, cancer and diverse neurological pathologies, are the consequence.
The discovery of the molecular principles of cellular transport was honored with the Nobel Prize of physiology and medicine in 2013. While knowing the intracellular roads and the functioning of the cars that use these roads is essential knowledge, one cannot understand much of how a cell functions without knowing how all this traffic is regulated and controlled.
Cell and systems biologists of the University of Zurich could now create a first global map of the regulatory control systems of the majority of transport routes in a cell. This unique map has been published by the renowned scientific journal Cell as its cover story.
How cells uptake and transport cargos
Cells regulate the uptake of nutrients and messenger cargos and their transport within the cell. This process is known as endocytosis and membrane traffic. Different cargos dock onto substrate specific receptors on the cell membrane. Special proteins such as kinases, GTPases and coats, activate specific entry routes and trigger the uptake of the receptors into the cell. For their uptake, the receptors and docked cargos become enclosed by the cell membrane. In the next steps, the membrane invaginates and becomes constricted. The resulting vesicle is guided via several distinct stations, cellular organelles, to its final destination in the cell.
Cells regulate the main routes, side routes and intersections
For her study, Dr. Prisca Liberali, senior scientist in the team of Professor Lucas Pelkmans, sequentially switched off 1200 human genes. Using automated high-throughput light microscopy and computer vision, she could monitor and compare 13 distinct transport paths involving distinct receptors and cellular organelles. Precise quantifications of thousands of single cells identified the genes required for the different transport routes. Surprisingly, sets of transport routes are co-regulated and coordinated in specific ways by different programs of regulatory control.
Subsequently, Dr. Liberali calculated the hierarchical order within the genetic network and thereby identified the regulatory topology of cellular transport. “The transport into the cell and within the cells proceeds analogously to the cargo transport within a city” describes the scientist. “Like in a city, the traffic on the routes within a cell and their intersections is tightly regulated by traffic lights and signs to guide the cargo flow.”
Thanks to this unique quantitative map, the fine regulatory details of transport paths and processes within a cells could be mapped for the first time. Particularly the genes that encode for these traffic lights and switches are often de-regulated in disease. With this map, it is now possible to predict how this leads to traffic jams in the cells, causing the disease phenotype. Alternatively, since many drugs have been developed to target these traffic lights and switches, the map can be used to come up with possible drug combinations to target unwanted traffic, such as viruses, to the waste disposal system of the cell.
Literature:
Prisca Liberali, Berend Snijder, Lucas Pelkmans. A hierarchical map of regulatory genetic interactions in membrane trafficking. Cell. June 5, 2014. DOI: http://dx.doi.org/10.1016/j.cell.2014.04.029
Contact:
Dr. Prisca Liberali
Institut für Molekulare Biologie
Universität Zürich
Tel. +41 44 635 31 94 / +41 77 459 57 53
E-Mail: prisca.liberali@imls.uzh.ch
Bettina Jakob
Media Relations
Universität Zürich
Tel. +41 44 634 44 39
E-Mail: bettina.jakob@kommunikation.uzh.ch
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















