Cellular defense against fatal associations between proteins and DNA
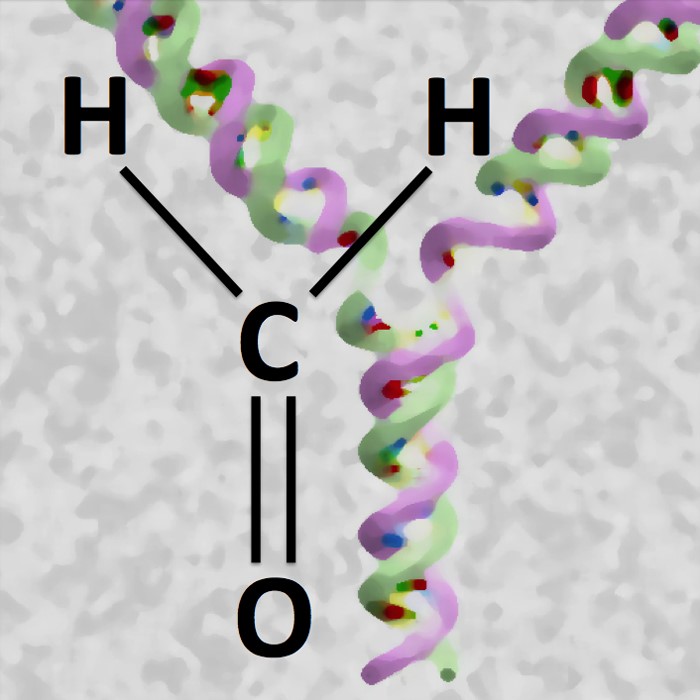
Formaldehyde can crosslink DNA to proteins, which interferes with DNA replication. The protein Wss1 chops down the protein component of these crosslinks, thus enabling cells to complete replication. Illustration: Stefan Jentsch / Copyright: MPI of Biochemistry
Very special DNA lesions are DNA-protein crosslinks (DPCs) – proteins covalently linked to DNA. So far hardly anything was known about repair mechanisms specifically targeting DPCs.
Stefan Jentsch’s team at the Max Planck Institute of Biochemistry (MPIB) in Martinsried, Germany, now discovered a protease that is able to chop down the protein component of DPCs, thereby enabling organisms to copy their genetic information even if DPCs arise. The results of this study have major implications for the understanding of genome integrity and cancer development.
The DNA in each cell is highly vulnerable to various types of damage. A special class of damage is caused by reactive compounds, such as formaldehyde, which are produced as byproducts of cellular reactions and cause the crosslinking (a formation of a covalent linkage) of proteins to DNA.
Importantly, these so-called DNA-protein crosslinks (DPCs) are also caused by several anti-cancer drugs and are extremely toxic as they interfere with essential processes such as DNA replication.
Cells need to unwind and separate the DNA double helix in order to copy its genetic information prior to the next round of cell division. DPCs inhibit this process by blocking the way of the unwinding enzyme (replicative helicase), thus preventing replication and consequently cell division.
In the laboratory of Stefan Jentsch at the MPIB, scientists now identified the protease Wss1 as a new safeguarding factor that chops down the protein components of DPCs and thereby enables cells to duplicate their genome. Julian Stingele, a PhD student in the laboratory, found that cells lacking Wss1 are particularly sensitive to formaldehyde, extremely vulnerable to DPCs and suffer from genomic instability.
Notably, Wss1 has the unique property to cleave proteins only in the presence of DNA, suggesting that the enzyme is well tailored for its task to remove DPCs from the genome and thus preserve genome stability.
Because the repair of DNA lesions is essential to prevent cancer formation, it is of crucial importance to understand the underlying cellular mechanisms. The newly identified DPC-repair pathway is particularly important for rapidly dividing cells. Given the fact that cancer cells divide much faster than the majority of human cells, Wss1 might be an attractive future drug target for cancer therapy.
Original Publication:
J. Stingele, M. Schwarz, N. Bloemeke, P. Wolf, and S. Jentsch: A DNA-dependent protease involved in DNA-protein crosslink repair. Cell, July 3, 2014.
DOI: 10.1016/j.cell.2014.04.053
Contact:
Prof. Dr. Stefan Jentsch
Molecular Cell Biology
Max Planck Institute of Biochemistry
Am Klopferspitz 18
82152 Martinsried
Germany
E-Mail: jentsch@biochem.mpg.de
http://www.biochem.mpg.de/jentsch
Anja Konschak
Public Relations
Max Planck Institute of Biochemistry
Am Klopferspitz 18
82152 Martinsried
Germany
Tel. +49 89 8578-2824
E-Mail: konschak@biochem.mpg.de
http://www.biochem.mpg.de/news
http://www.biochem.mpg.de/news/ueber_das_institut/forschungsbereiche/zellbiologi… – More Press Releases about the Research of Stefan Jentsch
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















