Cell contents may be key to controlling toxicity of Huntington's disease protein
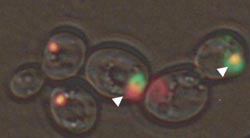
This is an image showing sequestration of the prion form of translation release factor Sup35 (red) by polyglutamines in an aggresome (green). A new study found that aggresome formation in yeast cells containing Sup35 did not reduce the toxicity of huntingtin proteins with a polyglutamine tract. The study suggests a potentially new approach for identifying possible therapeutic targets for treating Huntington’s disease. Credit: Georgia Tech/Yury Chernoff<br>
Huntington's disease causes the progressive breakdown of nerve cells in the brain and affects an individual's movement, cognition and mental state. Genetically, the disease is associated with a mutation in the Huntingtin gene that causes the huntingtin protein to be produced with an extended region containing the amino acid glutamine.
The mechanisms that control the severity and onset of the disease are poorly understood, as individuals with the same amount of expansion in their huntingtin proteins experience differences in toxicity and onset of the disease.
A new study led by Georgia Institute of Technology researchers suggests that the toxic effects of the huntingtin protein on cells may not be driven exclusively by the length of the protein's expansion, but also by which other proteins are present in the cell.
The researchers placed human huntingtin protein with an expanded region, called a polyglutamine tract, into yeast cells and found toxicity differences that were based on the other protein aggregates — called prions — present in the cells.
“This study clarifies genetic and epigenetic mechanisms that modulate polyglutamine's toxicity on cells and establishes a new approach for identifying potential therapeutic targets through characterization of pre-existing proteins in the cell,” said Yury Chernoff, a professor in the School of Biology at Georgia Tech. “While this study was conducted in yeast, it is possible that there are differences in aggregated proteins present in human cells as well, which are causing variation in huntingtin toxicity among individuals.”
The results of the study were published in the April 2012 issue of the journal PLoS Genetics. This work was supported by the National Institutes of Health and the Hereditary Disease Foundation.
Also contributing to this research were former Georgia Tech graduate student He Gong and postdoctoral fellow Nina Romanova, University of North Carolina at Chapel Hill School of Medicine research assistant professor Piotr Mieczkowski, and Boston University School of Medicine professor Michael Sherman.
Expanded huntingtin forms clumps in human cells that are typically transported and stored in an internal compartment called an aggresome until they can be removed from the body. While the compartment is thought to protect the contents of the cell from the toxic contents inside the aggresome, the current study shows that huntingtin molecules inside an aggresome can still be toxic to the cell.
In the study, aggresome formation in the cells containing the prion form of the Rnq1 protein reduced the toxicity of the huntingtin protein in Saccharomyces cerevisiae yeast cells, whereas the huntingtin protein's toxicity remained in the presence of the prion form of translation release factor Sup35.
“It remains uncertain whether the toxicity was primarily driven by sequestration of Sup35 into the aggresome or by its sequestration into the smaller huntingtin protein aggregates that remained in the cytoplasm,” explained Chernoff, who is also director of the Center for Nanobiology of the Macromolecular Assembly Disorders (NanoMAD). “While Sup35 was detected in the aggresome, we don't know if the functional fraction of Sup35 was sequestered there.”
In a follow-on experiment, the researchers increased the level of another release factor, Sup45, in the presence of Sup35 and found that this combination counteracted the toxicity.
“While the Rnq1 and Sup35 prions did not cause significant toxicity on their own, the results show that prion composition in the cell drove toxicity,” noted Chernoff. “Prions modulated which proteins were sequestered by the aggresome, as proteins associated with the pre-existing prions were more likely to be sequestered, such as Sup45 because of its association with Sup35.”
It remains unknown if polyglutamines can sequester the human versions of the Sup35 and Sup45 release factors, but this study shows the possibility that organisms may differ by the protein composition in their cells, and this in turn may influence their susceptibility to polyglutamine disorders such as Huntington's disease.
Research reported in this publication was supported by the National Institute of General Medical Sciences of the National Institutes of Health (NIH) under award numbers GM058763, GM093294 and GM086890. The content is solely the responsibility of the principal investigators and does not necessarily represent the official views of the NIH.
Media Contact
More Information:
http://www.gatech.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Machine learning algorithm reveals long-theorized glass phase in crystal
Scientists have found evidence of an elusive, glassy phase of matter that emerges when a crystal’s perfect internal pattern is disrupted. X-ray technology and machine learning converge to shed light…

Mapping plant functional diversity from space
HKU ecologists revolutionize ecosystem monitoring with novel field-satellite integration. An international team of researchers, led by Professor Jin WU from the School of Biological Sciences at The University of Hong…

Inverters with constant full load capability
…enable an increase in the performance of electric drives. Overheating components significantly limit the performance of drivetrains in electric vehicles. Inverters in particular are subject to a high thermal load,…





















