Appetite suppressant for scavenger cells
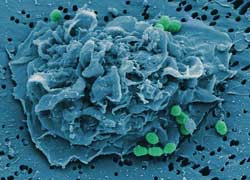
A scavenger cell of the immune system ingests bacteria (shown in green). During an influenza infection, the macrophages’ appetite is curbed.<br>Manfred Rohde/HZI<br>
When infected with influenza, the body becomes an easy target for bacteria. The flu virus alters the host’s immune system and compromises its capacity to effectively fight off bacterial infections. Now, a team of immunologists at the Helmholtz Centre for Infection Research (HZI) and cooperation partners has discovered that an immune system molecule called TLR7 is partly to blame. The molecule recognizes the viral genome – and then signals scavenger cells of the immune system to ingest fewer bacteria. The researchers published their findings in the Journal of Innate Immunity.
The flu is not just a seasonal illness during the winter months. In the past, there have been several flu pandemics that have claimed the lives of millions. By now, we know that during the course of the disease, many people not only get sick from the flu itself but also from bacterial pathogens like the much-feared pneumococci, the bacteria causing pneumonia.
In many cases, such “superinfections” can cause the disease to take a turn for the worse. In fact, during the Spanish Flu of 1918 to 1920, they were responsible for the majority of deaths. Why an infection with the flu virus increases the risk for superinfections is still poorly understood. Now, a group of scientists from HZI, the University Hospital of the Otto von Guericke University Magdeburg, the Essen University Hospital, the Karolinska Institute in Stockholm, Sweden, as well as further research institutions have discovered one more detail on how the virus manipulates the immune system.
They focused on TLR7, a molecule that is found in different cells of the body. TLR7 is capable of recognizing viral genetic material. As it turns out, TLR7 has an unwanted side effect, too: During a flu infection, it appears to undermine the body’s innate ability to fight off bacteria, thereby increasing the chance of a superinfection. The researchers made their discovery when they examined how superinfected mice were dealing with the bacterium Streptococcus pneumoniae, the pneumonia pathogen. The scientists colored the bacteria and measured how many of them were taken up by scavenger cells of the immune system called macrophages. The macrophages of TLR7-deficient mice had a bigger appetite and eliminated larger numbers of bacteria when infected with the flu than those of mice with the intact viral sensor. “Without TLR7, it takes longer before influenza-infected mice reach the critical point where they are no longer able to cope with the bacterial infection,” explains Prof. Dunja Bruder, head of HZI’s “Immune Regulation Group” and professor of infection immunology at the University Hospital Magdeburg.
The scientists also have an idea about how TLR7 may be curtailing the scavenger cells’ appetite: Whenever the immune system recognizes a virus, it gets other immune cells to produce a signaling substance called IFN gamma. It is already known that this substance inhibits macrophages in the lungs, causing them to eliminate fewer bacteria. As part of their study, the researchers discovered another indication of this special relationship: In TLR7-deficient animals they found smaller quantities of the IFN gamma messenger substance. The consequence might be that macrophages have a bigger appetite and that therefore bacterial entry into the bloodstream is delayed.
“Our results confirm that in the long run the flu virus suppresses the body’s ability to defend itself against bacteria. Presumably, this is an unwanted side effect of the viral infection,” speculates Dr. Stegemann-Koniszewski, the study’s first author.
“Unfortunately, it is rather difficult to intervene therapeutically. At first glance, it seems obvious to inhibit TLR7 during influenza so that the macrophages are actually able to get rid of the bacteria. However, this could have unforeseen repercussions as TLR7 and IFN gamma are both part of a tightly regulated immunological network,” explains Prof. Matthias Gunzer, former research group leader at the HZI and currently a professor at Essen University Hospital.
Even if a lack of TLR7 cannot by itself ward off a bacterial superinfection, the researchers’ findings could still lead to highly promising potential clinical applications. “Missing TLR7 delays the spread of bacteria via the bloodstream,” says Bruder. “Even if we are only talking about a relatively brief window of time, this might be our critical opportunity for keeping a seriously ill patient alive. The more time doctors have to choose the right antibiotic for their patient, the better the chances of a successful treatment.”
Original Publication
Sabine Stegemann-Koniszewski, Marcus Gereke, Sofia Orrskog, Stefan Lienenklaus, Bastian Pasche, Sophie R. Bader, Achim D. Gruber, Shizuo Akira, Siegfried Weiss, Birgitta Henriques-Normark, Dunja Bruder*, Matthias Gunzer* (* These authors contributed equally to the study.)
TLR7 contributes to the rapid progression but not to the overall fatal outcome of secondary pneumococcal disease following influenza A virus infection
Journal of Innate Immunity, 2012
DOI: 10.1159/000345112
The research group “Immune Regulation” at the HZI explores the immune system under extreme situations. These can be parallel infections with different pathogens or the erroneous attack of parts of the own body by the immune system.
The Helmholtz Centre for Infection Research (HZI):
The Helmholtz Centre for Infection Research contributes to the achievement of the goals of the Helmholtz Association of German Research Centres and to the successful implementation of the research strategy of the German Federal Government. The goal is to meet the challenges in infection research and make a contribution to public health with new strategies for the prevention and therapy of infectious diseases.
http://www.helmholtz-hzi.de
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Making diamonds at ambient pressure
Scientists develop novel liquid metal alloy system to synthesize diamond under moderate conditions. Did you know that 99% of synthetic diamonds are currently produced using high-pressure and high-temperature (HPHT) methods?[2]…

Eruption of mega-magnetic star lights up nearby galaxy
Thanks to ESA satellites, an international team including UNIGE researchers has detected a giant eruption coming from a magnetar, an extremely magnetic neutron star. While ESA’s satellite INTEGRAL was observing…

Solving the riddle of the sphingolipids in coronary artery disease
Weill Cornell Medicine investigators have uncovered a way to unleash in blood vessels the protective effects of a type of fat-related molecule known as a sphingolipid, suggesting a promising new…





















