Antibiotic resistance spreads rapidly between bacteria
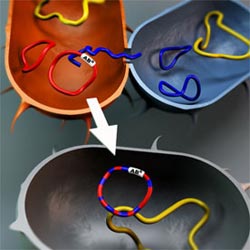
Antibiotic resistance-carrying plasmids from different bacteria can meet and exchange genetic material. The result is plasmids consisting of genes that have each been adapted to different bacterial species. This facilitates further adaptation and mobility, and consequently also the spread of antibiotic resistance between different bacterial species. Björn Norberg<br>
The results are published in an article in the scientific journal Nature Communications.
More and more bacteria are becoming resistant to our common antibiotics, and to make matters worse, more and more are becoming resistant to all known antibiotics. The problem is known as multi-resistance, and is generally described as one of the most significant future threats to public health Antibiotic resistance can arise in bacteria in our environment and in our bodies. Antibiotic resistance can then be transferred to the bacteria that cause human diseases, even if the bacteria are not related to each other.
A large proportion of gene transfer between bacteria takes place with the aid of what are known as conjugative plasmids, a part of the bacterial DNA. A plasmid can only exist and multiply inside a cell, where it uses the cell’s machinery, but can then be transferred to another cell and in that way spread between bacteria.
The research team has studied a group of the known carriers of antibiotic resistance genes: IncP-1 plasmids. Using advanced DNA analysis, the researchers have succeeded in mapping the origin of different IncP-1 plasmids and their mobility between different bacterial species. “Our results show that plasmids from the IncP-1 group have existed in, and adapted to, widely differing bacteria. They have also recombined, which means that a single plasmid can be regarded as a composite jigsaw puzzle of genes, each of which has adapted to different bacterial species”, says Peter Norberg, a researcher in the Institute of Biomedicine at the University of Gothenburg. This indicates very good adaptability and suggests that these plasmids can move relatively freely between, and thrive in, widely differing bacterial species.
“IncP-1 plasmids are very potent ‘vehicles’ for transporting antibiotic resistance genes between bacterial species. Therefore, it does not matter much in what environment, in what part of the world, or in what bacterial species antibiotic resistance arises. Resistance genes could relatively easily be transported from the original environment to bacteria that infect humans, through IncP-1 plasmids, or other plasmids with similar properties, as ‘vehicles’,” says Professor Malte Hermansson of the Department of Cell and Molecular Biology at the University of Gothenburg.
It has been known for some time that plasmids are important in the spread of antibiotic resistance. The research team’s findings show that IncP-1 plasmids can move, and have moved, between widely differing bacterial species and in addition have interacted directly with one another, which can increase the potential for gene spreading.
The study The IncP-1 Plasmid Backbone Adapts to Different Host Bacterial Species and Evolves Through Homologous Recombination has been conducted by Peter Norberg, Maria Bergström and Malte Hermansson at the University of Gothenburg, in cooperation with Vinay Jethava and Devdatt Dubhashi at the Chalmers University of Technology.
The research has been funded by the Swedish Research Council, the University of Gothenburg, the National Board of Health and Welfare, the Swedish Society of Medicine Funds, the Magnus Bergvall Foundation and the Wilhelm and Martina Lundgren Science Fund 1.
Contact:
Malte Hermansson, Department of Cell and Molecular Biology at the University of Gothenburg
+46 (0)31- 786 2574
malte.hermansson@cmb.gu.se
Peter Norberg, Institute of Biomedicine at the University of Gothenburg
+46 (0)735- 31 61 66
peter.norberg@gu.se
Bibiographic data
Journal: Nature Communiations
Title: The IncP-1 plasmid backbone adapts to different host bacterial species and evolves through homologous recombination
Authors: Peter Norberg, Maria Bergström, Vinay Jethava, Devdatt Dubhashi, Malte Hermansson
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















