A nucleotide change could initiate fragile X syndrome
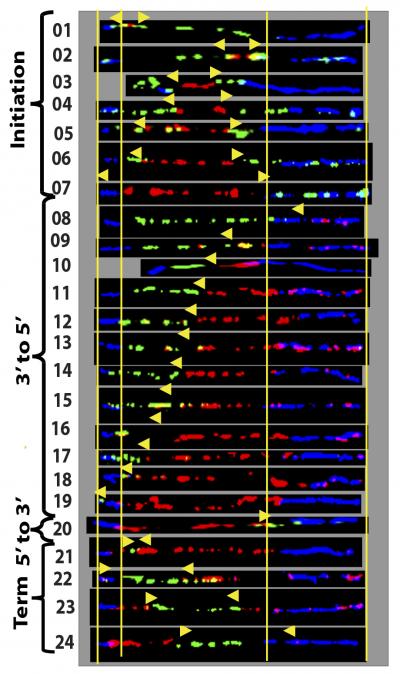
Researchers used genetic mapping to determine that stem cells derived from mothers carrying a fragile X premutation (above) show a normal pattern of DNA replication when a nearby DNA sequence is similar to normal stem cells. Credit: Gerhardt et al., 2014
Fragile X syndrome is caused by a defect in a gene on the X chromosome called fragile X mental retardation 1 (FMR1). Around 1 in 230 women and 1 in 360 men carry a so-called premutation, in which a series of DNA repeats at one end of the FMR1 gene is slightly longer than normal.
These repeats are prone to even further expansion when FMR1 is passed from mother to child, causing the gene to switch off and stop producing a protein that is important for some cognitive functions.
A group of researchers from Albert Einstein College of Medicine of Yeshiva University in New York previously found that a certain site that initiates DNA replication, located near to the FMR1 gene, is inactivated in fragile X embryonic stem cells.
This inactivation changes the way that the FMR1 gene is copied during cell division, which could pose problems that lead to expansion of the DNA repeats within the gene.
Intriguingly, a specific alteration in the DNA sequence near the FMR1 gene—a “single-nucleotide polymorphism” or SNP— has been linked to an increased risk of repeat expansion in some premutation carriers. These researchers discovered that this SNP overlaps with the inactive replication origin in fragile X embryonic stem cells.
Nucleotides in DNA include one of four bases (cytosine, thymine, adenine, or guanine). The researchers found that normal embryonic stem cells had a thymine base at the SNP site and an active replication origin. Fragile X cells, in contrast, had a cytosine base and an inactive origin.
The researchers also derived embryonic stem cells from mothers carrying the fragile X premutation. These cells had a thymine base and a normal replication pattern and, accordingly, showed no tendency to expand their repeat numbers over time.
The findings show that the substitution of cytosine for thymine might inactivate the DNA replication origin when the FMR1 gene is passed from mother to child, increasing the risk of DNA repeat expansions that can lead to fragile X syndrome.
Gerhardt, J., et al. 2014. J. Cell Biol. doi:10.1083/jcb.201404157
About The Journal of Cell Biology
The Journal of Cell Biology (JCB) is published by The Rockefeller University Press. All editorial decisions on manuscripts submitted are made by active scientists in conjunction with our in-house scientific editors. JCB content is posted to PubMed Central, where it is available to the public for free six months after publication. Authors retain copyright of their published works, and third parties may reuse the content for non-commercial purposes under a creative commons license. For more information, please visit http://www.jcb.org.
Research reported in this press release was supported by the National Institute of General Medical Sciences and National Institute of Child Health and Human Development of the National Institutes of Health, Empire State Stem Cell Fund, Starr Tri-Institutional Stem Cell Initiative, and Neurogenomics.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Silicon Carbide Innovation Alliance to drive industrial-scale semiconductor work
Known for its ability to withstand extreme environments and high voltages, silicon carbide (SiC) is a semiconducting material made up of silicon and carbon atoms arranged into crystals that is…

New SPECT/CT technique shows impressive biomarker identification
…offers increased access for prostate cancer patients. A novel SPECT/CT acquisition method can accurately detect radiopharmaceutical biodistribution in a convenient manner for prostate cancer patients, opening the door for more…

How 3D printers can give robots a soft touch
Soft skin coverings and touch sensors have emerged as a promising feature for robots that are both safer and more intuitive for human interaction, but they are expensive and difficult…





















