Rethinking the Basic Science of Graphene Synthesis
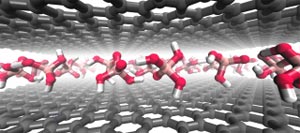
Mallouk Lab, Penn State Intercalation of graphite using Brønsted acids produces pristine single layer graphene.
In the decade since Nobel laureates Konstantin Novoselov and Andre Geim proved the remarkable electronic and mechanical properties of graphene, researchers have been hard at work to develop methods of producing pristine samples of the material on a scale with industrial potential. Now, a team of Penn State scientists has discovered a route to making single layer graphene that has been overlooked for more than 150 years.
“There are lots of layered materials similar to graphene with interesting properties, but until now we didn’t know how to chemically pull the solids apart to make single sheets without damaging the layers,” said Thomas E. Mallouk, Evan Pugh Professor of Chemistry, Physics, and Biochemistry and Molecular Biology at Penn State.
In a paper first published online Sept. 9 in the journal Nature Chemistry, Mallouk and colleagues at Penn State and the Research Center for Exotic Nanocarbons at Shinshu University, Japan, describe a method called intercalation, in which guest molecules or ions are inserted between the carbon layers of graphite to pull the single sheets apart.
The intercalation of graphite was achieved in 1841, but always with a strong oxidizing or reducing agent that damaged the desirable properties of the material. One of the mostly widely used methods to intercalate graphite by oxidation was developed in 1999 by Nina Kovtyukhova, a research associate in Mallouk’s lab.
While studying other layered materials, Mallouk asked Kovtyukhova to use her method, which requires a strong oxidizing agent and a mixture of acids, to open up single layers of solid boron nitride, a compound with a structure similar to graphene. To their surprise, she was able to get all of the layers to open up. In subsequent control experiments, Kovtyukhova tried leaving out various agents and found that the oxidizing agent wasn’t necessary for the reaction to take place.
Mallouk asked her to try a similar experiment without the oxidizing agent on graphite, but aware of the extensive literature saying that the oxidizing agent was required, Kovtyukhova balked.
“I kept asking her to try it and she kept saying no,” Mallouk said. “Finally, we made a bet, and to make it interesting I gave her odds. If the reaction didn’t work I would owe her $100, and if it did she would owe me $10. I have the ten dollar bill on my wall with a nice Post-it note from Nina complimenting my chemical intuition.”
Mallouk believes the results of this new understanding of intercalation in boron nitride and graphene could apply to many other layered materials of interest to researchers in the Penn State Center for Two-Dimensional and Layered Materials who are investigating what are referred to as “Materials Beyond Graphene.” The next step for Mallouk and colleagues will be to figure out how to speed the reaction up in order to scale up production.
Their results appear in the Nature Chemistry article titled “Non-oxidative intercalation and exfoliation of graphite by Brønsted acids” (http://dx.doi.org/10.1038/nchem.2054) by Nina I. Kovtyukhova, Yuanxi Wang, Ayse Berkdemir, Mauricio Terrones, Vincent H. Crespi and Thomas E. Mallouk, all of Penn State, and Rodolfo Cruz-Silva of the Research Center for Exotic Nanocarbons, Shinshu University, Nagano, Japan. Their work was supported by the U.S. Army Research Office MURI grant W911NF-11-1-0362. Contact Prof. Mallouk at tom@chem.psu.edu.
About the Center for Two Dimensional and Layered Materials at Penn State
The 2DLM Center conducts multidisciplinary research in the fast emerging field of atomically thin layered materials. Based in Penn State’s Materials Research Institute, the Center works with industry partners, national labs, and academic collaborators to discover and predict new properties that arise when novel materials are created one atomic layer at a time. Visit the website at http://www.mri.psu.edu/centers/2dlm
Contact Information
Walter Mills
Associate Editor Publications
wem12@psu.edu
Phone: 814-865-0285
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

Making diamonds at ambient pressure
Scientists develop novel liquid metal alloy system to synthesize diamond under moderate conditions. Did you know that 99% of synthetic diamonds are currently produced using high-pressure and high-temperature (HPHT) methods?[2]…

Eruption of mega-magnetic star lights up nearby galaxy
Thanks to ESA satellites, an international team including UNIGE researchers has detected a giant eruption coming from a magnetar, an extremely magnetic neutron star. While ESA’s satellite INTEGRAL was observing…

Solving the riddle of the sphingolipids in coronary artery disease
Weill Cornell Medicine investigators have uncovered a way to unleash in blood vessels the protective effects of a type of fat-related molecule known as a sphingolipid, suggesting a promising new…





















