Nanodiamands as bacterial killers
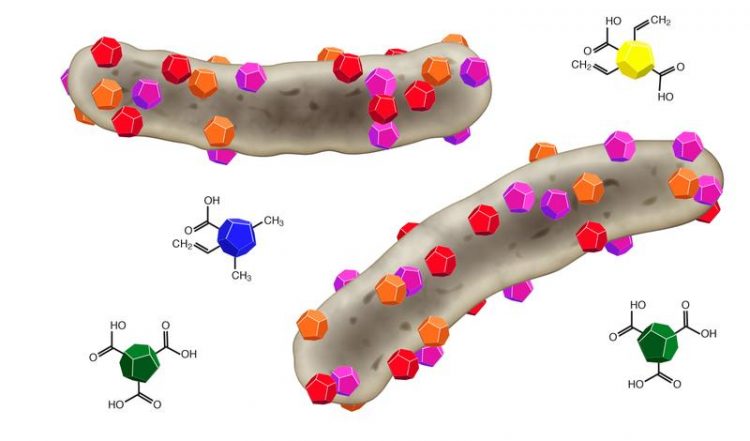
The colored particles display different types of nanodiamonds that bind to bacterial cells (grey) and kill them. Foto: Rezwan
Exhibiting a diameter of 5 nanometers, nanodiamonds are 200-times smaller than a bacterium. Nanodiamonds are produced by the explosion of carbon-containing compounds in high-pressure storage tanks. Here, the tiny detonation diamonds are formed besides large amounts of soot.
The material scientists Dr. Michael Maas, Julia Wehling and Professor Kurosch Rezwan from the University of Bremen (Germany) have now identified the strong antibacterial properties of these nanodiamonds. Besides silver and copper, nanodiamonds might be used as a new effective agent against bacterial contaminations and infections.
Discovered in the 1960s by Russian scientists, nanodiamonds only recently came into the spotlight, caused by current breakthroughs in processing and pretreatments that enabled their use in laboratories. Heat treatment of the grayish brown diamond powder can be used to generate different chemical groups on the nanodiamond surface. Biologist Julia Wehling and chemist and project leader Dr. Michael Maas discovered that some types of nanodiamonds kill bacterial cells rapidly and efficiently.
Seeking to understand the reason for the antibacterial properties, both material scientists from the Advanced Ceramics Group of Prof. Dr.-Ing. Kurosch Rezwan puzzled out the cause: some oxygen-containing groups on the surface of nanodiamonds, such as acid anhydrides, seem to be responsible for the antibacterial effect of the diamonds.
“The discovery that nanodiamonds kill bacterial cells as effectively as silver, which has been already used for 7000 years, opens a multitude of possible applications in biomedicine and material science. Furthermore, the concentrations that we used are proven to be nontoxic for human cells.
This enables the use of nanodiamonds for surface coatings or as additives for disinfectants. In the era of antibiotic resistances, the discovery of a new antibacterial material can be seen as a breakthrough”, says Julia Wehling.
The only scarcely explored diamonds were brought to the attention of Dr. Michael Maas by Prof. Richard N. Zare during a visit at Stanford University in California. “After my return, we directly started using nanodiamonds in the different nanosystems that we are working with in Bremen.
We were quite surprised by how efficiently nanodiamonds killed bacteria and we are convinced that our discovery will be of great impact for further research. It can be expected that nanodiamonds will play a key role in different areas dealing with bacterial infection. Our next goal is to equip implant materials with nanodiamonds to provide them with antibacterial properties. At the same time, we want to further analyze the diamond surface”, Michael Maas says.
Professor Kurosch Rezwan, director of the Advanced Ceramics department at the University of Bremen also sees great potential in the antibacterial nanodiamonds and points out that the publication in the renowned journal “ACS Nano” would not have been possible without the excellent collaboration with Prof. Dr. Ralf Dringen as the leader of the Biomolecular Interactions and Neurobiochemistry Group of the University of Bremen and Prof. Richard N. Zare of the department of chemistry of the Stanford University.
Full publication:
Julia Wehling, Ralf Dringen, Richard N. Zare, Michael Maas, Kurosch Rezwan: Bactericidal Activity of Partially Oxidized Nanodiamonds, ACS Nano, 2014, http://pubs.acs.org/doi/abs/10.1021/nn502230m.
Further information:
Universität Bremen
Fachbereich Produktionstechnik
Keramische Werkstoffe und Bauteile / Advanced Ceramics
M.Sc. Julia Wehling
Tel.: +49 421 218 64966
E-Mail: julia.wehling@uni-bremen.de
Dr. rer. nat. Michael Maas
Tel.: +49 421 218 64939
E-Mail: michael.maas@uni-bremen.de
Prof. Dr.-Ing. Kurosch Rezwan
Tel.: +49 421 218 64930
E-Mail: krezwan@uni-bremen.de
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…