Berkeley Lab Scientists Create ‘Molecular Paper’

Now, Berkeley Lab scientists have made the largest two-dimensional polymer crystal self-assembled in water to date. This entirely new material mirrors the structural complexity of biological systems with the durable architecture needed for membranes or integration into functional devices.
These self-assembling sheets are made of peptoids, engineered polymers that can flex and fold like proteins while maintaining the robustness of manmade materials. Each sheet is just two molecules thick yet hundreds of square micrometers in area—akin to ‘molecular paper’ large enough to be visible to the naked eye. What’s more, unlike a typical polymer, each building block in a peptoid nanosheet is encoded with structural ‘marching orders’—suggesting its properties can be precisely tailored to an application. For example, these nanosheets could be used to control the flow of molecules, or serve as a platform for chemical and biological detection.
“Our findings bridge the gap between natural biopolymers and their synthetic counterparts, which is a fundamental problem in nanoscience,” said Ronald Zuckermann, Director of the Biological Nanostructures Facility at the Molecular Foundry. “We can now translate fundamental sequence information from proteins to a non-natural polymer, which results in a robust synthetic nanomaterial with an atomically-defined structure.”
The building blocks for peptoid polymers are cheap, readily available and generate a high yield of product, providing a huge advantage over other synthesis techniques. Zuckermann, instrumental in developing the Foundry’s one-of-a-kind robotic synthesis capabilities, worked with his team of coauthors to form libraries of peptoid materials. After screening many candidates, the team landed upon the unique combination of polymer building blocks that spontaneously formed peptoid nanosheets in water.
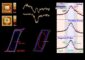
Zuckermann and coauthor Christian Kisielowski reached another first by using the TEAM 0.5 microscope at the National Center for Electron Microscopy (NCEM) to observe individual polymer chains within the peptoid material, confirming the precise ordering of these chains into sheets and their unprecedented stability while being bombarded with electrons during imaging.
“The design of nature-inspired, functional polymers that can be assembled into membranes of large lateral dimensions marks a new chapter for materials synthesis with direct impact on Berkeley Lab’s strategically relevant initiatives such as the Helios project or Carbon Cycle 2.0,” said NCEM’s Kisielowski. “The scientific possibilities that come with this achievement challenge our imagination, and will also help move electron microscopy toward direct imaging of soft materials.”
“This new material is a remarkable example of molecular biomimicry on many levels, and will no doubt lead to many applications in device fabrication, nanoscale synthesis and imaging,” Zuckermann added.
This research is reported in a paper titled, “Free floating ultra-thin two-dimensional crystals from sequence-specific peptoid polymers,” appearing in the journal Nature Materials and available in Nature Materials online. Co-authoring the paper with Zuckermann and Kisielowski were Ki Tae Nam, Sarah Shelby, Phillip Choi, Amanda Marciel, Ritchie Chen, Li Tan, Tammy Chu, Ryan Mesch, Byoung-Chul Lee and Michael Connolly.
This work at the Molecular Foundry was supported by DOE’s Office of Science and the Defense Threat Reduction Agency.
The Molecular Foundry is one of the five DOE Nanoscale Science Research Centers (NSRCs), premier national user facilities for interdisciplinary research at the nanoscale. Together the NSRCs comprise a suite of complementary facilities that provide researchers with state-of-the-art capabilities to fabricate, process, characterize and model nanoscale materials, and constitute the largest infrastructure investment of the National Nanotechnology Initiative. The NSRCs are located at DOE’s Argonne, Brookhaven, Lawrence Berkeley, Oak Ridge and Sandia and Los Alamos National Laboratories. For more information about the DOE NSRCs, please visit http://nano.energy.gov.
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California. Visit our website at http://www.lbl.gov.
Additional Information
For more about Berkeley Lab’s Molecular Foundry visit http://foundry.lbl.gov/
For more about the DOE NSRCs visit http://nano.energy.gov
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

“Nanostitches” enable lighter and tougher composite materials
In research that may lead to next-generation airplanes and spacecraft, MIT engineers used carbon nanotubes to prevent cracking in multilayered composites. To save on fuel and reduce aircraft emissions, engineers…

Trash to treasure
Researchers turn metal waste into catalyst for hydrogen. Scientists have found a way to transform metal waste into a highly efficient catalyst to make hydrogen from water, a discovery that…

Real-time detection of infectious disease viruses
… by searching for molecular fingerprinting. A research team consisting of Professor Kyoung-Duck Park and Taeyoung Moon and Huitae Joo, PhD candidates, from the Department of Physics at Pohang University…